$\gamma = \frac{{{C_P}}}{{{C_V}}} = \frac{5}{3}$
we know $\Delta Q = n{C_P}\Delta T$ and $\Delta U = n{C_V}\Delta T$
==> $\frac{{\Delta U}}{{\Delta Q}} = \frac{{{C_V}}}{{{C_P}}} = \frac{3}{5}$
i.e. fraction of heat energy to increase the internal energy be $3/5.$
Download our appand get started for free
Similar Questions
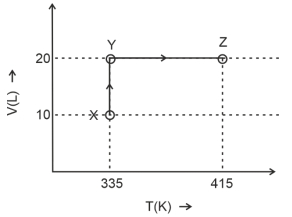
- 1Consider the following volume-temperature $( V - T )$ diagram for the expansion of $5$ moles of an ideal monoatomic gas.View Solution
Considering only $P-V$ work is involved, the total change in enthalpy (in Joule) for the transformation of state in the sequence $X \rightarrow Y \rightarrow Z$ is $\qquad$
[Use the given data: Molar heat capacity of the gas for the given temperature range, $C _{ v , m }=12 J K ^{-1} mol ^{-1}$ and gas constant, $R =8.3 J K ^{-1} mol ^{-1}$ ]
- 2A mixture of ideal gas containing $5$ moles of monatomic gas and $1$ mole of rigid diatomic gas is initially at pressure $P _0$, volume $V _0$ and temperature $T _0$. If the gas mixture is adiabatically compressed to a volume $V _0 / 4$, then the correct statement(s) is/are,View Solution
(Give $2^{1.2}=2.3 ; 2^{3.2}=9.2 ; R$ is gas constant)
$(1)$ The final pressure of the gas mixture after compression is in between $9 P _0$ and $10 P _0$
$(2)$ The average kinetic energy of the gas mixture after compression is in between $18 RT _0$ and $19 RT _0$
$(3)$ The work $| W |$ done during the process is $13 RT _0$
$(4)$ Adiabatic constant of the gas mixture is $1.6$
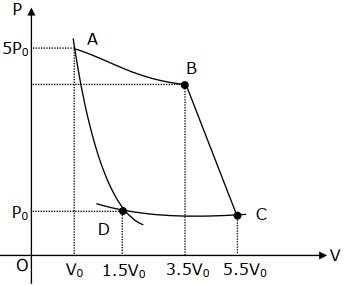
- 3In the reported figure, there is a cyclic process $ABCDA$ on a sample of $1\, {mol}$ of a diatomic gas. The temperature of the gas during the process ${A} \rightarrow {B}$ and ${C} \rightarrow {D}$ are ${T}_{1}$ and ${T}_{2}\left({T}_{1}\,>\,{T}_{2}\right)$ respectively.View Solution
Choose the correct option out of the following for work done if processes $B C$ and $D A$ are adiabatic.
- 4An ideal refrigerator has a freezer at a temperature of $ - 13^\circ C.$ The coefficient of performance of the engine is $5.$ The temperature of the air (to which heat is rejected) will beView Solution
- 5View SolutionA gas undergoes a change at constant temperature. Which of the following quantities remain fixed?
- 6A Carnot engine with efficiency $50\,\%$ takes heat from a source at $600\,K$. In order to increase the efficiency to $70\,\%$, keeping the temperature of sink same, the new temperature of the source will be $.........\,K$View Solution
- 7A gas ($\gamma = 1.3)$ is enclosed in an insulated vessel fitted with insulating piston at a pressure of ${10^5}\,N/{m^2}$. On suddenly pressing the piston the volume is reduced to half the initial volume. The final pressure of the gas isView Solution
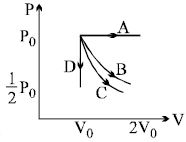
- 8The $PV$ diagram shows four different possible reversible processes performed on a monatomic ideal gas. Process $A$ is isobaric (constant pressure). Process $B$ is isothermal (constant temperature). Process $C$ is adiabatic. Process $D$ is isochoric (constant volume). For which process(es) does the temperature of the gas decrease ?View Solution
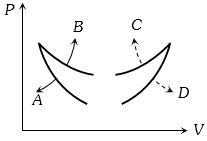
- 9Four curves $A, B, C$ and $D$ are drawn in the adjoining figure for a given amount of gas. The curves which represent adiabatic and isothermal changes areView Solution
- 10If the temperature of sink is at absolute zero, then the efficiency of Carnot engine will be ........ $\%$View Solution
