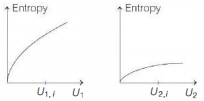
Graphs below show the entropy versus energy $U$ of two systems $1$ and $2$ at constant volume. The initial energies of the systems are indicated by $U_{1, i}$ and $U_{2, i}$, respectively. Graphs are drawn to the same scale. The systems are then brought into thermal contact with each other. Assume that, at all times the combined energy of the two systems remains constant. Choose the most appropriate option indicating the energies of the two systems and the total entropy after they achieve the equilibrium.

KVPY 2018, Advanced
$(c)$ By second law of thermodynamics, entropy of a system either increases or remains constant (only in reversible quasistatic processes).
Also, from graphs given
$S_{2} > S_{1} \Rightarrow \frac{Q}{T_{2}} > \frac{Q}{T_{1}}$
where, $S$ is entropy, $Q$ is heat and $T$ is the temperature.
or $\quad T_{1} < T_{2}$
Hence, temperature of system $1$ is less than system $2$. So, heat flows from system $2$ to system $1$ .
Hence, $U_{1}$ increases and $U_{2}$ decreases.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The value of $\eta$ may lie betweenView Solution
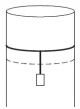
- 2A long cylindrical pipe of radius $20 \,cm$ is closed at its upper end and has an airtight piston of negligible mass as shown. When a $50 \,kg$ mass is attached to the other end of the piston, it moves down. If the air in the enclosure is cooled from temperature $T$ to $T-\Delta T$, the piston moves back to its original position. Then $\Delta T / T$ is close to (Assuming air to be an ideal gas, $g=10 \,m / s ^2$, atmospheric pressure is $10^5 \,Pa$ )View Solution
- 3View SolutionA gas is compressed adiabatically, which one of the following statement is NOT true.
- 4View SolutionThe internal energy of the gas increases In
- 5Heat energy of $735\,J$ is given to a diatomic gas allowing the gas to expand at constant pressure. Each gas molecule rotates around an internal axis but do not oscillate. The increase in the internal energy of the gas will be $..........\,J$View Solution
- 6$N$ moles of an ideal diatomic gas are in a cylinder at temperature $T$. suppose on supplying heat to the gas, its temperature remain constant but $n$ moles get dissociated into atoms. Heat supplied to the gas isView Solution
- 7A monoatomic gas of $n-$moles is heated from temperature $T_1$ to $T_2$ under two different conditions $(i)$ at constant volume and $(ii)$ at constant pressure. The change in internal energy of the gas isView Solution
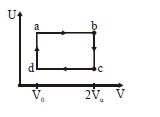
- 8The figure given below shows the variation in the internal energy $U$ with volume $V$ of $2.0\ mole$ of an ideal gas in a cyclic process $abcda$ . The temperatures of the gas during the processes $ab$ and $cd$ are $500\ K$ and $300\ K$ respectively, the heat absorbed by the gas during the complete process is .... $J$View Solution
(Take $R$ = $8.3\ J/mol-K$ and $ln\ 2$ = $0.69$)
- 9One mole of an ideal gas at initial temperature $T$, undergoes a quasi-static process during which the volume $V$ is doubled. During the process, the internal energy $U$ obeys the equation $U=a V^3$, where $a$ is a constant. The work done during this process isView Solution
- 10A Car not engine whose low temperate reservoir is at $7\,^oC$ has an efficiency of $50\%$ . It is desired to increase the efficiency to $70\%$ . By how many degrees should the temperature of the high temperature reservoir be increased .... $K$View Solution
