Download our appand get started for free
Similar Questions
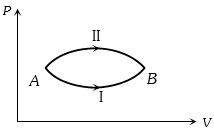
- 1A system goes from $A$ to $B$ via two processes $I$ and $II$ as shown in figure. If $\Delta {U_1}$ and $\Delta {U_2}$ are the changes in internal energies in the processes $I$ and $II$ respectively, thenView Solution
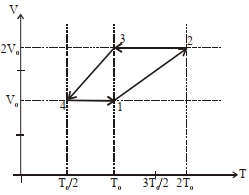
- 2One mole of a monoatomic ideal gas goes through a thermodynamic cycle, as shown in the volume versus temperature ($V-T$) diagram. The correct statement($s$) is/are :View Solution
[ $R$ is the gas constant]
$(1)$ Work done in this thermodynamic cycle $(1 \rightarrow 2 \rightarrow 3 \rightarrow 4 \rightarrow 1)$ is $| W |=\frac{1}{2} RT _0$
$(2)$ The ratio of heat transfer during processes $1 \rightarrow 2$ and $2 \rightarrow 3$ is $\left|\frac{ Q _{1 \rightarrow 2}}{ Q _{2 \rightarrow 3}}\right|=\frac{5}{3}$
$(3)$ The above thermodynamic cycle exhibits only isochoric and adiabatic processes.
$(4)$ The ratio of heat transfer during processes $1 \rightarrow 2$ and $3 \rightarrow 4$ is $\left|\frac{Q_{U \rightarrow 2}}{Q_{3 \rightarrow 4}}\right|=\frac{1}{2}$
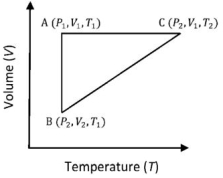
- 3A reversible cyclic process for an ideal gas is shown below. Here, $P, V$, and $T$ are pressure, volume and temperature, respectively. The thermodynamic parameters $q, w, H$ and $U$ are heat, work, enthalpy and internal energy, respectively.View Solution
(image)
The correct option ($s$) is (are)
$(A)$ $q_{A C}=\Delta U_{B C}$ and $W_{A B}=P_2\left(V_2-V_1\right)$
$(B)$ $\mathrm{W}_{\mathrm{BC}}=\mathrm{P}_2\left(\mathrm{~V}_2-\mathrm{V}_1\right)$ and $\mathrm{q}_{\mathrm{BC}}=\mathrm{H}_{\mathrm{AC}}$
$(C)$ $\Delta \mathrm{H}_{\mathrm{CA}}<\Delta \mathrm{U}_{\mathrm{CA}}$ and $\mathrm{q}_{\mathrm{AC}}=\Delta \mathrm{U}_{\mathrm{BC}}$
$(D)$ $\mathrm{q}_{\mathrm{BC}}=\Delta \mathrm{H}_{\mathrm{AC}}$ and $\Delta \mathrm{H}_{\mathrm{CA}}>\Delta \mathrm{U}_{\mathrm{CA}}$
- 4The temperature of reservoir of Carnot's engine operating with an efficiency of $70\%$ is $1000K.$ The temperature of its sink is ...... $K$View Solution
- 5An ideal gas is taken through a quasi-static process described by $P = \alpha\, V^2$, with $\alpha = 5\,atm/m^6$. The gas is expanded to twice its original volume of $1\,m^3$. How much work is done by the expanding gas in this processView Solution
- 6One mole of ${O_2}$ gas having a volume equal to $22.4$ litres at ${0^o}C$ and $1$ atmospheric pressure in compressed isothermally so that its volume reduces to $11.2$ litres. The work done in this process is ...... $J$View Solution
- 7When $1\, gm$ of water at ${0^o}C$ and $1 \times {10^5}\;N/{m^2}$ pressure is converted into ice of volume $1.091\;c{m^2}$, the external work done will beView Solution
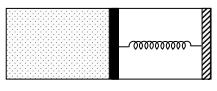
- 8An ideal monoatomic gas is confined in a horizontal cylinder by a spring loaded piston (as shown in the figure). Initially the gas is at temperature $T _1$, pressure $P_1$ and volume $V_1$ and the spring is in its relaxed state. The gas is then heated very slowly to temperature $T_2$, pressure $P _2$ and volume $V _2$. During this process the piston moves out by a distance $x$. Ignoring the friction between the piston and the cylinder, the correct statement$(s)$ is(are)View Solution
$(A)$ If $V_2=2 V_1$ and $T_2=3 T_1$, then the energy stored in the spring is $\frac{1}{4} P_1 V_1$
$(B)$ If $V_2=2 V_1$ and $T_2=3 T_1$, then the change in internal energy is $3 P_1 V_1$
$(C)$ If $V_2=3 V_1$ and $T_2=4 T_1$, then the work done by the gas is $\frac{7}{3} P_1 V_1$
$(D)$ If $V_2=3 V_1$ and $T_2=4 T_1$, then the heat supplied to the gas is $\frac{17}{6} P_1 V_1$
- 9One mole of a monatomic ideal gas undergoes an adiabatic expansion in which its volume becomes eight times its initial value. If the initial temperature of the gas is $100 K$ and the universal gas constant $R =8.0 Jmol ^{-1} K ^{-1}$, the decrease in its internal energy, in Joule, is. . . . .View Solution
- 10View SolutionIn practice, all heat engines have efficiency less than that of a Carnot engine because
