If $\Delta Q$ and $\Delta W$ represent the heat supplied to the system and the work done on the system respectively, then the first law of thermodynamics can be written as
AIIMS 2013, Easy
From FLOT $\Delta Q=\Delta U+\Delta W$
$\because$ Heat supplied to the system so
$\Delta Q \rightarrow\, Positive$
and work is done on the system so
$\Delta \mathrm{W} \rightarrow\,Negative$
Hence $+\Delta Q=\Delta U-\Delta W$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A Carnot engine with sink's temperature at $17\,^oC$ has $50\%$ efficiency. By how much should its source temperature be changed to increases its efficiency to $60\%$ ?...... $K$View Solution
- 2Under isothermal condition, the pressure of a gas is given by $P = aV ^{-3}$, where $a$ is a constant and $V$ is the volume of the gas. The bulk modulus at constant temperature is equal to $..........\,P$View Solution
- 3A certain amount of gas is taken through a cyclic process $(A\,B\,C\,D\,A)$ that has two isobars, one isochore and one isothermal. The cycle can be represented on a $P-V$ indicator diagram asView Solution
- 4A Carnot engine absorbs an amount $Q$ of heat from a reservoir at an absolute temperature $T$ and rejects heat to a sink at a temperature of $T/3$ . The amount of heat rejected isView Solution
- 5View SolutionThe work done in which of the following processes is zero
- 6A thermo-dynamical system is changed from state $({P_1},\,{V_1})$ to $({P_2},\,{V_2})$ by two different process. The quantity which will remain same will beView Solution
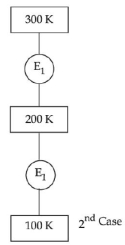
- 7In $1^{\text {st }}$ case, Carnot engine operates between temperatures $300\,K$ and $100\,K$. In $2^{\text {nd }}$ case, as shown in the figure, a combination of two engines is used. The efficiency of this combination (in $2^{\text {ad }}$ case) will be.View Solution
- 8An ideal gas expands in such a manner that its pressure and volume can be related by equation $P{V^2} = $ constant. During this process, the gas isView Solution
- 9If the amount of heat given to a system be $35$ joules and the amount of work done by the system be $ - 15$ joules, then the change in the internal energy of the system is .... $joules$View Solution
- 10Consider a spherical shell of radius $R$ at temperature $T$. The black body radiation inside it can be considered as an ideal gas of photons with internal energy per unit volume$E=$ $\frac{U}{V} \propto {T^4}$ and pressure $P = \frac{1}{3}\left( {\frac{U}{V}} \right)$ If the shell now undergoes an adiabatic expansion the relation between $T$ and $R$ isView Solution
