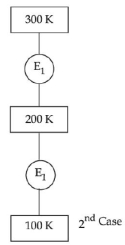
In $1^{\text {st }}$ case, Carnot engine operates between temperatures $300\,K$ and $100\,K$. In $2^{\text {nd }}$ case, as shown in the figure, a combination of two engines is used. The efficiency of this combination (in $2^{\text {ad }}$ case) will be.

JEE MAIN 2022, Medium
First case : $\eta=1-\frac{100}{300}=\frac{2}{3}$
Second case : $\eta_{\text {net }}=\eta_{1}+\eta_{2}-\eta_{1} \eta_{2}$
$\eta_{1}=1-\frac{200}{300}=\frac{1}{3}$
$\eta_{2}=1-\frac{100}{200}=\frac{1}{2}$
$\eta_{\text {net }}=\frac{1}{3}+\frac{1}{2}-\frac{1}{6}=\frac{2}{3}$
$\eta$ (first case) $=\eta$ (second case)
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionIn isothermal expansion, the pressure is determined by
- 2In an isothermal reversible expansion, if the volume of $96\, gm$ of oxygen at $27°C$ is increased from $70$ litres to $140$ litres, then the work done by the gas will beView Solution
- 3In a thermodynamic process, pressure of a fixed mass of a gas is changed in such a manner that the gas molecules gives out $20 J$ of heat and $10 J$ of work is done on the gas. If the initial internal energy of the gas was $40 J,$ then the final internal energy will be ........ $J$View Solution
- 4View SolutionOut of the following which quantity does not depend on path
- 5$1\,g$ of a liquid is converted to vapour at $3 \times 10^5\,Pa$ pressure. If $10 \%$ of the heat supplied is used for increasing the volume by $1600\,cm ^3$ during this phase change, then the increase in internal energy in the process will be $............\,J$View Solution
- 6Two Carnot engines $A$ and $B$ are operated in series. The first one, $A,$ receives heat at $T_1(= 600\,K)$ and rejects to a reservoir at temperature $T_2.$ The second engine $B$ receives heat rejected by the first engine and, in turns, rejects to a heat reservoir at $T_3 (=400\,K).$ Calculate the temperature $T_2$ if the work outputs of the two engines are equal ..... $K$View Solution
- 7Air is pumped into a balloon, of initial volume $V$ , until its diameter has doubled. If the atmospheric pressure is $p$ , what is the work done against the atmosphere ?View Solution
- 8Match the thermodynamic processes taking place in a system with the correct conditions. In the table: $\Delta Q$ is the heat supplied, $\Delta W$ is the work done and $\Delta U$ is change in internal energy of the systemView Solution
Process Condition $(I)$ Adiabatic $(A)\; \Delta W =0$ $(II)$ Isothermal $(B)\; \Delta Q=0$ $(III)$ Isochoric $(C)\; \Delta U \neq 0, \Delta W \neq 0 \Delta Q \neq 0$ $(IV)$ Isobaric $(D)\; \Delta U =0$ - 9A Carnot engine works between $27^{\circ} C$ and $127^{\circ} C$. Heat supplied by the source is $500\, J$. then heat ejected to the sink is (in $J$)View Solution
- 10View SolutionIn the following indicator diagram, the net amount of work done will be