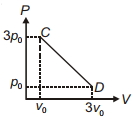
In the following indicator diagram, the net amount of work done will be
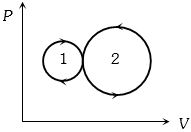

Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A closed vessel contains $0.1$ mole of a monoatomic ideal gas at $200\, K$. If $0.05$ mole of the same gas at $400\, K$ is added to it, the final equilibrium temperature (in $K$ ) of the gas in the vessel will be closed toView Solution
- 2One mole of an ideal gas $(\gamma = 1.4)$ is adiabatically compressed so that its temperature rises from $27\,^oC$ to $35\,^oC$ . The change in the internal energy of the gas is .... $J$. (given $R = 8.3\,J/mole-K$ )View Solution
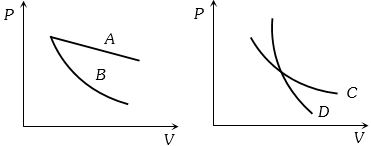
- 3In the following figure, four curves $A, B, C$ and $D$ are shown. The curves areView Solution
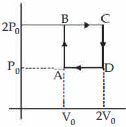
- 4Helium gas goes through a cycle $ABCDA$ ( consisting of two isochoric and isobaric lines) as shown in figure Efficiency of this cycle is nearly ....... $\%$ (Assume the gas to be close to ideal gas)View Solution
- 5View SolutionIn isothermal expansion, the pressure is determined by
- 6Two Carnot engines $A$ and $B$ are operated in succession. The first one, $A$ receives heat from a source at $T_1 = 800\, K$ and rejects to sink at $T_2K$. The second engine $B$ receives heat rejected by the first engine and rejects to another sink at $T_3 = 300\, K$. If the work outputs of two engines are equal, then the value of $T_2$ is ...... $K$View Solution
- 7A thermodynamic system goes from states $(i)\,\,{P_1}, V$ to $2{P_1}, V\, (ii)\, P, V$ to $P, 2V.$ Then work done in the two cases isView Solution
- 8The coefficient of performance of a refrigerator is $5.$ If the temperature inside freezer is $-20^o C,$ the temperature of the surroundings to which it rejects heat is ........ $^oC$View Solution
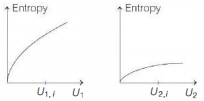
- 9Graphs below show the entropy versus energy $U$ of two systems $1$ and $2$ at constant volume. The initial energies of the systems are indicated by $U_{1, i}$ and $U_{2, i}$, respectively. Graphs are drawn to the same scale. The systems are then brought into thermal contact with each other. Assume that, at all times the combined energy of the two systems remains constant. Choose the most appropriate option indicating the energies of the two systems and the total entropy after they achieve the equilibrium.View Solution
- 10The process $C D$ is shown in the diagram. As system is taken from $C$ to $D$, what happens to the temperature of the system?View Solution