Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A sample of an ideal gas is taken through the cyclic process $abca$ as shown in the figure. The change in the internal energy of the gas along the path $ca$ is $-180\, J$. The gas absorbs $250\, J$ of heat along the path $ab$ and $60\, J$ along the path $bc$. The work done by the gas along the path $abc$ is ..... $J$View Solution
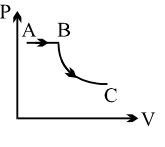
- 2An ideal gas is taken through a quasi-static process described by $P = \alpha\, V^2$, with $\alpha = 5\,atm/m^6$. The gas is expanded to twice its original volume of $1\,m^3$. How much work is done by the expanding gas in this processView Solution
- 3A Carnot freezer takes heat from water at $0\,^oC$ inside it and rejects it to the room at a temperature of $27\,^oC$. The latent heat of ice is $336 \times 10^3\, J\,kg^{-1}$. lf $5\, kg$ of water at $0\,^oC$ is converted into ice at $0\,^oC$ by the freezer, then the energy consumed by the freezer is close toView Solution
- 4A refrigerator is to maintain eatables kept inside at $9^{\circ} C .$ If room temperature is $36^{\circ} C$ calculate the coefficient of performance.View Solution
- 5$1\, mole$ of an ideal monoatomic gas at temperature $'T_0'$ expands slowly according to the law $P = KV$, where $K$ is a constant. If the final temperature of the gas is $2T_0$. Find the heat supplied to the gasView Solution
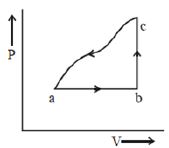
- 6A sample of ideal monoatomic gas is taken round the cycle $ABCA$ as shown in the figure. The work done during the cycle isView Solution
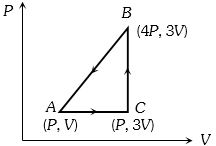
- 7One mole of a monoatomic ideal gas $\left(c_{ V }=\frac{3}{2} R \right)$ undergoes a cycle where it first goes isochorically from the state $\left(\frac{3}{2} P _0, V _0\right)$ to $\left( P _0, V _0\right)$, and then is isobarically contracted to the volume $\frac{1}{2} V _0$. It is then taken back to the initial state by a path which is a quarter ellipse on the $P - V$ diagram. The efficiency of this cycle isView Solution
- 8$PV$ curve for the process whose $VT$ curve isView Solution
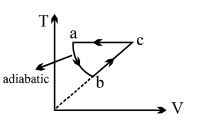
- 9The pressure in the tyre of a car is four times the atmospheric pressure at $300 K$. If this tyre suddenly bursts, its new temperature will be $(\gamma = 1.4)$View Solution
- 10View SolutionA process is shown in the diagram. Which of the following curves may represent the same process ?