A refrigerator is to maintain eatables kept inside at $9^{\circ} C .$ If room temperature is $36^{\circ} C$ calculate the coefficient of performance.
Easy
Temperature inside the refrigerator, $T_{1}=9^{\circ} C =282 K$
Room temperature, $T_{2}=36^{\circ} C =309 K$
Coefficient of performance $=\frac{T_{1}}{T_{2}-T_{1}}$
$=\frac{282}{309-282}$
$=10.44$
Therefore, the coefficient of performance of the given refrigerator is $10.44$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1One mole of an ideal gas goes from an initial state $A$ to final state $B$ via two processes : It first undergoes isothermal expansion from volume $V$ to $3\, V$ and then its volume is reduced from $3\, V$ to $V$ at constant pressure. The correct $P-V$ diagram representing the two processes isView Solution
- 2One mole of an ideal gas with $\gamma = 1.4$, is adiabatically compressed so that its temperature rises from $27°C$ to $35°C$. The change in the internal energy of the gas is ....... $J$ $(R = 8.3\,J/mol.K)$View Solution
- 3One mole of an ideal monoatomic gas is heated at a constant pressure of one atmosphere from ${0^o}C$ to ${100^o}C$. Then the change in the internal energy isView Solution
- 4View SolutionA given system undergoes a change in which the work done by the system equals the decrease in its internal energy. The system must have undergone an
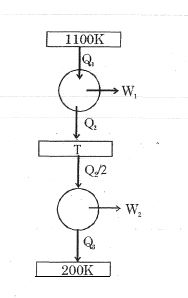
- 5Consider the given series combination of carnot cycles. If $W_1 = W_2$ then the value of $T$ is ...... $K$ (all temperatures are maintained at their respective values)View Solution
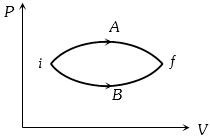
- 6In the figure given two processes $A$ and $B$ are shown by which a thermo-dynamical system goes from initial to final state $F.$ If $\Delta {Q_A}$ and $\Delta {Q_B}$ are respectively the heats supplied to the systems thenView Solution
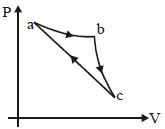
- 7A Carnot's engine working between $400\, K$ and $800\, K$ has a work output of $1200\, J$ per cycle. The amount of heat energy supplied to the engine from the source in each cycle is ........... $J$View Solution
- 8View SolutionPressure versus temperature graph of an ideal gas is shown in figure
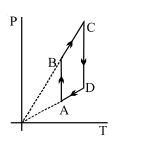
- 9An ideal gas at pressure $P$ and volume $V$ is expanded to volume$ 2V.$ Column $I$ represents the thermodynamic processes used during expansion. Column $II$ represents the work during these processes in the random order.:View Solution
Column $I$ Column $II$ $(p)$ isobaric $(x)$ $\frac{{PV(1 - {2^{1 - \gamma }})}}{{\gamma - 1}}$ $(q)$ isothermal $(y)$ $PV$ $(r)$ adiabatic (z) $PV\,\iota n\,2$ The correct matching of column $I$ and column $II$ is given by
- 10In the $P-V$ diagram shown, the gas does $5\, J$ of work in isothermal process $ab$ and $4\,J$ in adiabatic process $bc$. .... $J$ will be the change in internal energy of the gas in straight path $c$ to $a$ ?View Solution