In case of an adiabatic process the correct relation in terms of pressure $p$ and density $\rho $ of a gas is
Medium
Note that $p V^{\gamma}=$ constant and that $\rho$ is inversely
proportional to $V.$
Adiabatic process
$\mathrm{pV}^{\gamma}=$ constant
$\mathrm{p}\left(\frac{\mathrm{M}}{\rho}\right)^{\gamma}=\mathrm{constant}$
$\mathrm{p} \cdot \mathrm{M}^{\gamma} \cdot \rho^{-\gamma}=$ constant $\quad\left(\mathrm{M}^{\gamma} \text { is constant }\right)$
$\mathrm{p} \cdot \rho^{-\gamma}=$ constant
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionA gas undergoes a change at constant temperature. Which of the following quantities remain fixed?
- 2A Carnot engine operates between ${227^o}C$ and ${27^o}C.$ Efficiency of the engine will beView Solution
- 3The heat energy required to raise the temperature of $5\,moles$ of an ideal gas to $5\,K$ at constant pressure is $600\,J$ . How much heat (in $J$ ) is required to raise the same mass of the same gas to $5\,K$ at constant volume ? (Take $R = 8.3\,J/mole-^oK$ )View Solution
- 4One mole of an ideal gas undergoes a cyclic process, consisting of two isochores and two isobars. Temperature at $1$ and $3$ equal to $T_1$ and $T_3$ respectively. The work done by the gas over the cycle, if the point $2$ and $4$ lie on the same isothermView Solution
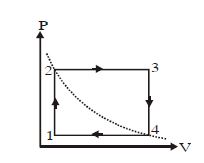
- 5A cyclic process $ABCA$ is shown in $PT$ diagram. When presented on $PV$, it wouldView Solution
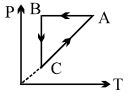
- 6Heat given to a system is $35$ joules and work done by the system is $15$ joules. The change in the internal energy of the system will be ..... $J$View Solution
- 7Find the amount of work done to increase the temperature of one mole of an ideal gas by $30^o\ C$ if it is expanding under the condition .... $J$View Solution
$V\propto {T^{\frac{2}{3}}}$ $[R = 1.99\ cal/mol-K]$
- 8A thermodynamic process is the pressure and volumes corresponding to some points in the figure are, $P_A = 3 \times 10^4 Pa$, $V_A = 2 \times 10^{-3}\, m^3$, $P_B = 8 \times 10^4 Pa$, $V_D = 5 \times 10^{-3}\,m^3$. In process $AB, 600\, J$ of heat and in process $BC, 200\, J$ of heat is added to the system. The change in the internal energy in process $AC$ would be .... $J$View Solution
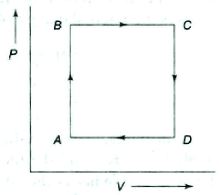
- 9An ideal gas expands in such a manner that its pressure and volume can be related by equation $P{V^2} = $ constant. During this process, the gas isView Solution
- 10The coefficient of performance of a refrigerator is $5.$ If the temperature inside freezer is $-20^o C,$ the temperature of the surroundings to which it rejects heat is ........ $^oC$View Solution
