The heat energy required to raise the temperature of $5\,moles$ of an ideal gas to $5\,K$ at constant pressure is $600\,J$ . How much heat (in $J$ ) is required to raise the same mass of the same gas to $5\,K$ at constant volume ? (Take $R = 8.3\,J/mole-^oK$ )
Diffcult
At constant pressure, heat energy required is given by
$\mathrm{Q}_{\mathrm{p}}=\mu \mathrm{C}_{\mathrm{p}} \Delta \mathrm{T}=600 \mathrm{J}(\text { given })$
where $\mu$ is the number of moles of ideal gas. At constant volume,
$\mathrm{Q}_{\mathrm{v}}=\mu \mathrm{C}_{\mathrm{v}} \mathrm{dT}=\mu\left(\mathrm{C}_{\mathrm{p}}-\mathrm{R}\right) \Delta \mathrm{T}$
$\left(\because C_{p}-C_{v}=R\right)$
$=600-\mu R . \Delta T$
$=600-5 \times 8.31 \times 5$
$=600-207.75=392.25 \mathrm{J}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A cyclic process for $1\,mole$ of an ideal gas is shown. Find work done in $AB, BC$ and $CA$ respectivelyView Solution
- 2View SolutionA process can be reversible if
- 3An ideal gas is expanding such that $\mathrm{PT}^2=$ constant. The coefficient of volume expansion of the gas isView Solution
- 4View SolutionA process can be reversible if
- 5In a process, temperature and volume of one mole of an ideal monoatomic gas are varied according to the relation $VT = K$, where $I$ is a constant. In this process the temperature of the gas is increased by $\Delta T$. The amount of heat absorbed by gas is ($R$ is gas constant)View Solution
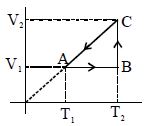
- 6Two moles of helium gas are taken over the cycle $ABCDA$, as shown in the $P-T$ diagram. Assuming the gas to be ideal the work done on the gas in taking it from $A $ to $B$ is ...... $R$View Solution

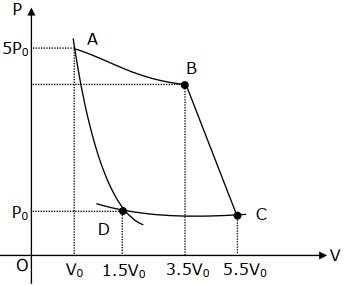
- 7In the reported figure, there is a cyclic process $ABCDA$ on a sample of $1\, {mol}$ of a diatomic gas. The temperature of the gas during the process ${A} \rightarrow {B}$ and ${C} \rightarrow {D}$ are ${T}_{1}$ and ${T}_{2}\left({T}_{1}\,>\,{T}_{2}\right)$ respectively.View Solution
Choose the correct option out of the following for work done if processes $B C$ and $D A$ are adiabatic.
- 8One mole of an ideal gas at temperature $T_1$ expends according to the law $\frac{P}{{{V^2}}} =a$ (constant). The work done by the gas till temperature of gas becomes $T_2 $ isView Solution
- 9A gas is compressed from a volume of $2\,m^3$ to a volume of $1\, m^3$ at a constant pressure of $100\, N/m^2$. Then it is heated at constant volume by supplying $150\, J$ of energy. As a result, the internal energy of the gasView Solution
- 10A sample of gas expands from $V_1$ to $V _2$. In which of the following, the work done will be greatest ?View Solution