In Newton's experiment of cooling, the water equivalent of two similar calorimeters is $10 $ gm each. They are filled with $350 gm$ of water and $300 gm$ of a liquid (equal volumes) separately. The time taken by water and liquid to cool from ${70^o}C$ to ${60^o}C$ is $3$ min and $95$ sec respectively. The specific heat of the liquid will be ...... $Cal/gm\,^oC$
Easy
(c) ${S_l} = \frac{1}{{{m_l}}}\left[ {\frac{{{t_l}}}{{{t_W}}}({m_W}{C_W} + W) - W} \right]$
$ = \frac{1}{{300}}\left[ {\frac{{95}}{{3 \times 60}}(350 \times 1 + 10) - 10} \right]$ $=0.6 Cal/gm\times°C$
$ = \frac{1}{{300}}\left[ {\frac{{95}}{{3 \times 60}}(350 \times 1 + 10) - 10} \right]$ $=0.6 Cal/gm\times°C$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionHeat travels through vacuum by
- 2If a liquid takes $30 \;sec$ in cooling from $80^{\circ} C$ to $70^{\circ} C$ and $70 \;sec$ in cooling from $60^{\circ} C$ to $50^{\circ} C$, then find the room temperature.View Solution
- 3Two metallic spheres ${S_1}$ and ${S_2}$are made of the same material and have identical surface finish. The mass of ${S_1}$ is three times that of ${S_2}$. Both the spheres are heated to the same high temperature and placed in the same room having lower temperature but are thermally insulated from each other. The ratio of the initial rate of cooling of ${S_1}$ to that of ${S_2}$ isView Solution
- 4View SolutionA thermos flask is polished well
- 5A rod $C D$ of thermal resistance $10.0\; {KW}^{-1}$ is joined at the middle of an identical rod ${AB}$ as shown in figure, The end $A, B$ and $D$ are maintained at $200^{\circ} {C}, 100^{\circ} {C}$ and $125^{\circ} {C}$ respectively. The heat current in ${CD}$ is ${P}$ watt. The value of ${P}$ is ... .View Solution
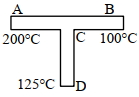
- 6The initial temperature of a body is $80°C$ . If its temperature falls to $64°C$ in $5$ minutes and in $10$ minutes to $52°C$ then the temperature of surrounding will be ...... $^oC$View Solution
- 7Surface of the lake is at $2°C$ . Find the temperature of the bottom of the lake..... $^oC$View Solution
- 8Two cylinders $P$ and $Q$ have the same length and diameter and are made of different materials having thermal conductivities in the ratio $2 : 3$ . These two cylinders are combined to make a cylinder. One end of $P$ is kept at $100°C$ and another end of $Q$ at $0°C$ . The temperature at the interface of $P$ and $Q$ is ...... $^oC$View Solution
- 9Three very large plates of same area are kept parallel and close to each other. They are considered as ideal black surfaces and have very high thermal conductivity. The first and third plates are maintained at temperatures $2T$ and $3T$ respectively. The temperature of the middle (i.e. second) plate under steady state condition isView Solution
- 10A slab of stone of area $0.36\;m ^2$ and thickness $0.1 \;m$ is exposed on the lower surface to steam at $100^{\circ} C$. A block of ice at $0^{\circ} C$ rests on the upper surface of the slab. In one hour $4.8\; kg$ of ice is melted. The thermal conductivity of slab is .......... $J / m / s /{ }^{\circ} C$ (Given latent heat of fusion of ice $=3.36 \times 10^5\; J kg ^{-1}$)View Solution
