Lower alcohols are highly soluble in water due to _____.
- H-bonding
The solubility of lower alcohols is due to the existence of hydrogen bonds between water and polar -OH group of alcohol molecules.
The -OH group in alcohols and phenols contain a hydrogen bonded to an electronegative oxygen atom. Thus they form hydrogen bonds with water molecules.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$\text{CH}_3\text{−CH}\text{−CH}−\text{CH}_3\\ \ \ \ \ \ \ \ \ \ \ \ \ | \ \ \ \ \ \ \ \ \ \ | \\ \ \ \ \ \ \ \ \ \ \text{OH}\ \ \ \ \ \ \text{OH}$IUPAC name of the given compound is:View Solution
- 2View SolutionWhich of the following can not be formed by hydrolysis of alkenes?
- 3$........$ is prepared on a large seals from water gas.View Solution
- 4View SolutionWhich of the following alcohols is the least soluble in water?
- 5View SolutionArrange the following alkanols 1, 2 and 3 in order of their reactivity towards acid catalyzed dehydration.
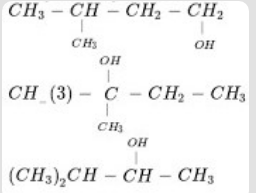
- 6What is the $\text{IUPAC}$ name of the following compound? $\ce{CH_3−CH_2−CH_2−OH}$View Solution
- 7View SolutionWhich of the following alcohol has highest solubility in water?
- 8In iodine charge transfer test the color of ether compound formed is$:$View Solution
- 9When alkyl halide is heated with dry $\ce{Ag_2O,}$ it produces:View Solution
- 10View SolutionWhich of the following alcohol has the highest boiling point?
