Step $1$ It is first compressed adiabatically from volume $V_{1}$ to $1 \;m ^{3}$.
Step $2$ Then expanded isothermally to volume $10 \;m ^{3}$.
Step $3$ Then expanded adiabatically to volume $V _{3}$.
Step $4$ Then compressed isothermally to volume $V_{1}$. If the efficiency of the above cycle is $3 / 4$, then $V_{1}$ is ............ $m^3$
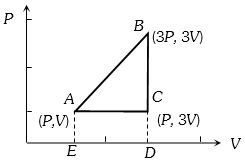
$A B:$ Adiabatic compression,
$V_{A}=V_{1}, V_{B}=1 \,m ^{3}$
$B C:$ Isothermal expansion,
$V_{C}=V=10 \,m ^{3}$
$C D$ : Adiabatic expansion,
$V_{D}=V_{3}$
$D A$ : Isothermal compression,
$V_{A}=V_{1}$
Cycle efficiency is given, $\eta=\frac{3}{4}$
For given Carnot's cycle,
$\eta=1-\frac{T_{1}}{T_{2}}=1-\left(\frac{V_{2}}{V_{1}}\right)^{\gamma-1}$
$[\therefore$ Process $A B$ is adiabatic, $\gamma=\frac{5}{3}$ for monoatomic gas]
$\Rightarrow \quad \frac{3}{4}=1-\left(\frac{1}{V_{1}}\right)^{\frac{5}{3}-1} \Rightarrow \frac{1}{V_{1}^{2 / 3}}=\frac{1}{4}$
$\Rightarrow V_{1}=8 \,m ^{3}$
Download our appand get started for free
Similar Questions
- 1A sample of $0.1\, g$ of water at $100^o C$ and normal pressure $(1.013 \times 10^5 N m^{-2} )$ requires $54\ cal $ of heat energy to convert to steam at $100^o C.$ If the volume of the steam produced is $167.1 \,cc,$ the change in internal energy of the sample, is ....... $J$View Solution
- 2An engine operating between the boiling and freezing points of water will haveView Solution
$1.$ efficiency more than $27 \%$
$2.$ efficiency less than the efficiency a Carnot engine operating between the same two temperatures.
$3.$ efficiency equal to $27 \%$
$4.$ efficiency less than $27 \%$
- 3If the amount of heat given to a system be $35$ joules and the amount of work done by the system be $ - 15$ joules, then the change in the internal energy of the system is .... $joules$View Solution
- 4A heat engine is involved with exchange of heat of $1915\, J,$ $-40\, J ,+125\, J$ and $-Q\,J$, during one cycle achieving an efficiency of $50.0 \%$. The value of $Q$ is.......$J$View Solution
- 5The process $AB$ is shown in the diagram. As the gas is taken from $A$ to $B$, its temperatureView Solution
- 6A bicycle tyre is filled with air having pressure of $270\,kPa$ at $27^{\circ}\,C$. The approximate pressure of the air in the tyre when the temperature increases to $36^{\circ}\,C$ is $............kPa$View Solution
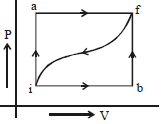
- 7When a system is taken from a state $i$ to $f$ along the path $iaf$ (as shown in the figure). $Q = 50\, cal$ and $W = 20\, cal$ ; along path $ibf,$ $Q = 36\, cal.$View Solution
$(i)$ What is $W$ along path $ibf$ ?
$(ii)$ If $W = 13$ cal for path $fi$, what is $Q$ for the path $fi$ ?
$(iii)$ Take $E_{int,i} = 10\,\, cal$ then what is $E_{int,f}$ ?
- 8View SolutionIf an ideal gas is compressed isothermally then
- 9An ideal gas at atmospheric pressure is adiabatically compressed so that its density becomes $32$ times of its initial value. If the final pressure of gas is $128$ atmosphers, the value of $\gamma$ the gas isView Solution
- 10An ideal gas is taken around $ABCA$ as shown in the above $P-V$ diagram. The work done during a cycle isView Solution