Starting at temperature $300\; \mathrm{K},$ one mole of an ideal diatomic gas $(\gamma=1.4)$ is first compressed adiabatically from volume $\mathrm{V}_{1}$ to $\mathrm{V}_{2}=\frac{\mathrm{V}_{1}}{16} .$ It is then allowed to expand isobarically to volume $2 \mathrm{V}_{2} \cdot$ If all the processes are the quasi-static then the final temperature of the gas (in $\left. \mathrm{K}\right)$ is (to the nearest integer)
JEE MAIN 2020, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$Assertion :$ The heat supplied to a system is always equal to the increase in its internal energy.View Solution
$Reason :$ When a system changes from one thermal equilibrium to another, some heat is absorbed by it. - 2Thermodynamic process is shown below on a $P-V$ diagram for one mole of an ideal gas. If $V _{2}=2 V _{1}$ then the ratio of temperature $T _{2} / T _{1}$ is ...... .View Solution
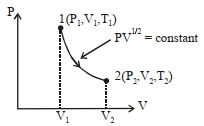
- 3The temperature inside a refrigerator is $t_2 \,^o C$ and the room temperature is $t_1\,^o C.$ The amount of heat delivered to the room for each joule of electrical energy consumed ideally will beView Solution
- 4One mole of a monatomic ideal gas undergoes an adiabatic expansion in which its volume becomes eight times its initial value. If the initial temperature of the gas is $100 K$ and the universal gas constant $R =8.0 Jmol ^{-1} K ^{-1}$, the decrease in its internal energy, in Joule, is. . . . .View Solution
- 5Unit mass of a liquid with volume ${V_1}$ is completely changed into a gas of volume ${V_2}$ at a constant external pressure $P$ and temperature $T.$ If the latent heat of evaporation for the given mass is $L,$ then the increase in the internal energy of the system isView Solution
- 6A cyclic process $ABCA$ is shown in $PT$ diagram. When presented on $PV$, it wouldView Solution
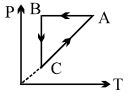
- 7A bicycle tyre is filled with air having pressure of $270\,kPa$ at $27^{\circ}\,C$. The approximate pressure of the air in the tyre when the temperature increases to $36^{\circ}\,C$ is $............kPa$View Solution
- 8View SolutionTwo gases are said to be in thermal equilibrium when they have same
- 9View SolutionWork done on or by a gas, in general depends upon the
- 10The coefficient of performance of a Carnot refrigerator working between ${30^o}C$ and ${0^o}C$ isView Solution
