One mole of a monatomic ideal gas undergoes an adiabatic expansion in which its volume becomes eight times its initial value. If the initial temperature of the gas is $100 K$ and the universal gas constant $R =8.0 Jmol ^{-1} K ^{-1}$, the decrease in its internal energy, in Joule, is. . . . .
IIT 2018, Medium
Adiabatic Expansion
$PV ^\gamma=\text { constant }$
$T . V ^{\gamma-1}=\text { constant }$
$T _2= T _1\left(\frac{ V _1}{ V _2}\right)^{\gamma-1} \text { where } \gamma=\frac{5}{3}$
$T _2= T _1\left(\frac{1}{8}\right)^{2 / 3}=\frac{ T _1}{4}=25 K$
$\Delta U =\frac{ f }{2} nR \Delta T =\frac{3}{2} \times 1 \times 8 \times 75=900 J$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$Assertion :$ Thermodynamic process in nature are irreversible.View Solution
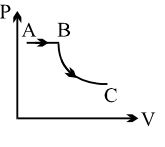
$Reason :$ Dissipative effects cannot be eliminated. - 2The pressure of a gas changes linearly with volume from $A$ to $B$ as shown in figure. If no heat is supplied to or extracted from the gas then change in the internal energy of the gas will be $............\,J$View Solution
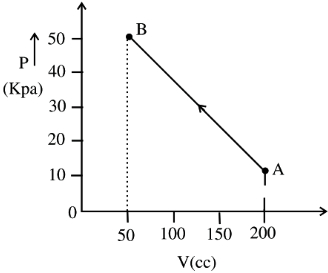
- 3A system is given $300$ calories of heat and it does $600$ joules of work. How much does the internal energy of the system change in this process? $( J =4.18$ $Joules / cal )$ (in joule)View Solution
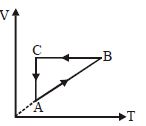
- 4A cyclic process $ABCA$ is shown in the $V-T$ diagram. Process on the $P-V$ diagram isView Solution
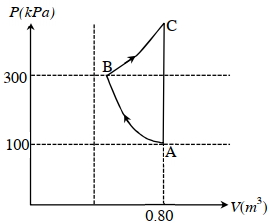
- 5In the given $P$-V diagram, a monoatomic gas $\left(\gamma=\frac{5}{3}\right)$ is first compressed adiabatically from state $A$ to state $B$. Then it expands isothermally from state $B$ to state $C$. [Given: $\left(\frac{1}{3}\right)^{0.6} \simeq 0.5, \ln 2 \simeq 0.7$ ].View Solution
Which of the following statement($s$) is(are) correct?
$(A)$ The magnitude of the total work done in the process $A \rightarrow B \rightarrow C$ is $144 kJ$.
$(B)$ The magnitude of the work done in the process $B \rightarrow C$ is $84 kJ$.
$(C)$ The magnitude of the work done in the process $A \rightarrow B$ is $60 kJ$.
$(D)$ The magnitude of the work done in the process $C \rightarrow A$ is zero.
- 6View SolutionA process can be reversible if
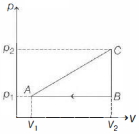
- 7An ideal gas with constant heat capacity $C_V=\frac{3}{2} n R$ is made to carry out a cycle that is depicted by a triangle in the figure given below. The following statement is true about the cycle.View Solution
- 8View SolutionIn thermodynamics, heat and work are
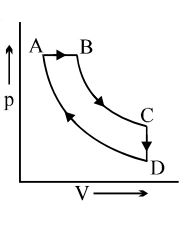
- 9A cyclic process $ABCD$ is shown in the $p-V$ diagram. Which of the following curves represents the same process if $BC \& DA$ are isothermal processesView Solution
- 10View SolutionA process is shown in the diagram. Which of the following curves may represent the same process ?