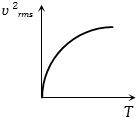
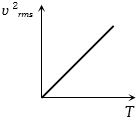
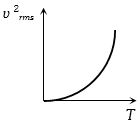
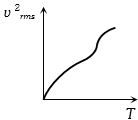
The curve between absolute temperature and ${v^2}_{rms}$ is
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1What is the velocity of wave in monoatomic gas having pressure $1$ kilopascal and density $2.6\,kg/{m^3}$View Solution
- 2According to the kinetic theory of gases the $r.m.s.$ velocity of gas molecules is directly proportional toView Solution
- 3If temperature of gas increases from $27°C$ to $927°C$ the $K.E.$ will beView Solution
- 4A cylinder of fixed capacity $44.8$ litre. contains a monatomic gas at standard temperature and pressure. The amount of heat required to cylinder by $10^o C$ will be. ($R=$ universal gas constant)View Solution
- 5View SolutionFor an ideal gas
- 6$Assertion :$ At a given temperature the specific heat of a gas at constant pressure. is always greater than its specific heat at constant volumeView Solution
$Reason :$ When a gas is heated at constant volume some extra heat is needed compared to that at constant pressure for doing work in expansion.
- 7During an adiabatic process, the pressure of a gas is found to be proportional to the cube of its absolute temperature. The ratio of $\frac{C_p}{C_v}$ for the gas is:View Solution
- 8The average translational kinetic energy of ${N}_{2}$ gas molecules at $\ldots \ldots \ldots . .{ }^{\circ} {C}$ becomes equal to the ${K} . {E}$. of an electron accelerated from rest through a potential difference of $0.1$ $volt.$View Solution
$\left(\right.$ Given $\left.{k}_{{B}}=1.38 \times 10^{-23} \, {J} / {K}\right)$
(Fill the nearest integer).
- 9Two non-reactive monoatomic ideal gases have their atomic masses in the ratio $2: 3$. The ratio of their partial pressures, when enclosed in a vessel kept at a constant temperature, is $4: 3$. The ratio of their densities is:View Solution
- 10A container of fixed volume has a mixture of one mole of hydrogen and one mole of helium in equilibrium at temperature $T$. Assuming the gases are ideal, the correct statement$(s)$ is(are)View Solution
$(A)$ The average energy per mole of the gas mixture is $2RT$.
$(B)$ The ratio of speed of sound in the gas mixture to that in helium gas is $\sqrt{6 / 5}$.
$(C)$ The ratio of the rms speed of helium atoms to that of hydrogen molecules is $1 / 2$.
$(D)$ The ratio of the rms speed of helium atoms to that of hydrogen molecules is $1 / \sqrt{2}$.