The metal capable of gaining as well as losing an electron is:
- Au
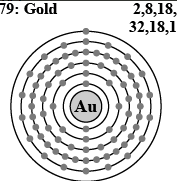
As shown in image, Gold has 2 electrons in level 1, 8 electrons in level 2, 18 in level 3, 32 in level 4, 18 in level 5 and 1 electron in level 6. Gold is having 1 eletcron in last level, so it can lost 1 electron to complete their valency or it can also gain 1 electron thus 2 electron will come in outer cell and will complete the last level.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionWhich elements shows the highest number of oxidation states ?
- 2View SolutionAmong the following metals, interatomic forces are probably the weakest in:
- 3View SolutionCatalytic activity of transition elements and their compounds is due to their _______.
- 4Lanthanides and actinides resemble in$:$View Solution
- 5View SolutionMetallic radii of some transition elements are given below. Which of these elements will have highest density?
Element Fe Co Ni Cu Metallic radii/pm 126 125 125 128 - 6View SolutionInterstitial compounds are formed when small atoms are trapped inside the crystal lattice of metals. Which of the following is not the characteristic property of interstitial compounds?
- 7View SolutionWhich of the following is true regarding derivation of the name of californium?
- 8View SolutionThe Lanthanoid contraction refers to:
- 9Although $+3$ is the characteristic oxidation state for lanthanoids but cerium also shows $+4$ oxidation state because $.......$View Solution
- 10When $\ce{KMnO_4}$ solution is added to oxalic acid solution, the decolourisation is slow in the beginning but becomes instantaneous after some time becauseView Solution
