The number of molecules in one litre of an ideal gas at $300 \,{K}$ and $2$ atmospheric pressure with mean kinetic energy $2 \times 10^{-9}\, {J}$ per molecules is $....\, \times 10^{11}$
JEE MAIN 2021, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$Assertion :$ For a gas atom the number of degrees of freedom is $3$.View Solution
$Reason :$ $\frac{{{C_P}}}{{{C_V}}} = \gamma $ - 2If the molecular weight of two gases are $M_1$ and $ M_1$, then at a temperature the ratio of root mean square velocity $v_1$ and $v_2$ will beView Solution
- 3A cylinder contains $10\, kg$ of gas at pressure of ${10^7}\,\,N/{m^2}.$ The quantity of gas taken out of the cylinder, if final pressure is $2.5 \times {10^6}\,N/{m^2},$ will be ..... $kg$ (Temperature of gas is constant)View Solution
- 4A vessel contains a mixture of one mole of oxygen and two moles of nitrogen at $300\ K.$ The ratio of the average rotational kinetic energy per $O_2$ molecule to that per $N_2$ molecule isView Solution
- 5View SolutionIt is possible for a substance to coexist in all three phases in equilibrium, when the substance is at
- 6View SolutionMoon has no atmosphere because
- 7Two spherical vessel of equal volume, are connected by a n arrow tube. The apparatus contains an ideal gas at one atmosphere and $300K$. Now if one vessel is immersed in a bath of constant temperature $600K$ and the other in a bath of constant temperature $300K$. Then the common pressure will be ...... $atm$View Solution
- 8At what temperature volume of an ideal gas at $0°C$ becomes triple ....... $^oC$View Solution
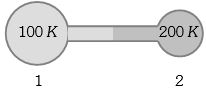
- 9Figure shows two flasks connected to each other. The volume of the flask $1$ is twice that of flask $2.$ The system is filled with an ideal gas at temperature $100\, K$ and $200 \,K $ respectively. If the mass of the gas in $1$ be $m$ then what is the mass of the gas in flask $2$View Solution
- 10Two vessels $A$ and $B$ are of the same size and are at same temperature. A contains $1 \mathrm{~g}$ of hydrogen and $B$ contains $1 \mathrm{~g}$ of oxygen. $\mathrm{P}_{\mathrm{A}}$ and $\mathrm{P}_{\mathrm{B}}$ are the pressures of the gases in $A$ and $\mathrm{B}$ respectively, then $\frac{\mathrm{P}_{\mathrm{A}}}{\mathrm{P}_{\mathrm{B}}}$ is:View Solution
