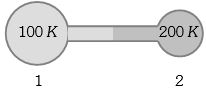
Figure shows two flasks connected to each other. The volume of the flask $1$ is twice that of flask $2.$ The system is filled with an ideal gas at temperature $100\, K$ and $200 \,K $ respectively. If the mass of the gas in $1$ be $m$ then what is the mass of the gas in flask $2$

Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1On $0^\circ C$ pressure measured by barometer is $760\, mm.$ What will be pressure on $100^\circ C$View Solution
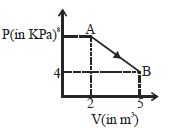
- 2A mole of an ideal diatomic gas undergoes a transition from $A$ to $B$ along a path $AB$ as shown in the figure, The change in internal energy of the gas during the transition is ...... $kJ$View Solution
- 3An ideal gas undergoes a quasi static, reversible process in which its molar heat capacity $C$ remains constant. If during this process the relation of pressure $P$ and volume $V$ is given by $PV^n =$ constant, then n is given by (Here $C_p$ and $C_v$ are molar specific heat at constant pressure and constant volume, respectively) :View Solution
- 4Hydrogen gas is filled in a balloon at $20^\circ C$. If temperature is made $40^\circ C$, pressure remaining same, what fraction of Hydrogen will come outView Solution
- 5When $x$ amount of heat is given to a gas at constant pressure, it performs $x/3$ amount of work. The average number of degrees of freedom per molecule isView Solution
- 6The pressure and volume of saturated water vapour are $P$ and $V$ respectively. It is compressed isothermally thereby volume becomes $V/2,$ the final pressure will beView Solution
- 7View SolutionWhich of the following statement is true
- 8Nitrogen gas is filled in an insulated container. If $\alpha$ fraction of moles dissociates without exchange of any energy, then the fractional change in its temperature is ..............View Solution
- 9View SolutionBoyle's law holds for an ideal gas during
- 10If number of molecules of ${H_2}$ are double than that of ${O_2}$, then ratio of kinetic energy of hydrogen and that of oxygen at $300 \,K$ isView Solution
