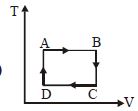
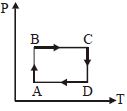
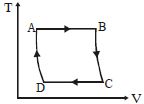
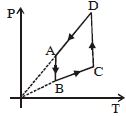
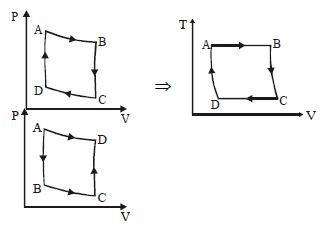
The $P-V$ diagram of a certain process (carnot cycle) is as shown. The process is also represent as
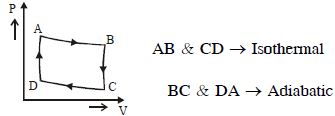
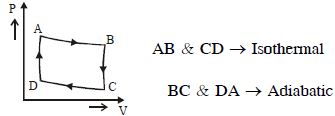
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionAn ideal gas undergoes a thermodynamics cycle as shown in figure. Which of the following graphs represents the same cycle?
- 2$Assertion :$ When a glass of hot milk is placed in a room and allowed to cool, its entropy decreases.View Solution
$Reason :$ Allowing hot object to cool does not violate the second law of thermodynamics. - 3Consider two containers $A$ and $B$ containing monoatomic gases at the same Pressure $(P)$, Volume $(V)$ and Temperature $(T)$. The gas in $A$ is compressed isothermally to $\frac{1}{8}$ of its original volume while the gas $B$ is compressed adiabatically to $\frac{1}{8}$ of its original volume. The ratio of final pressure of gas in $B$ to that of gas in $A$ is ...........View Solution
- 4Graph $A-B$ is an adiabatic curve. Choose the correct statementView Solution
- 5A gas expands with temperature according to the relation $V = k{T^{2/3}}.$ What is the work done when the temperature changes by ${30^o}C$View Solution
- 6In a process, temperature and volume of one mole of an ideal monoatomic gas are varied according to the relation $VT = K$, where $I$ is a constant. In this process the temperature of the gas is increased by $\Delta T$. The amount of heat absorbed by gas is ($R$ is gas constant)View Solution
- 7If the amount of heat given to a system be $35$ joules and the amount of work done by the system be $ - 15$ joules, then the change in the internal energy of the system is .... $joules$View Solution
- 8An engineer claims to have made an engine delivering $10 kW$ power with fuel consumption of $1 g/sec$. The calorific value of the fuel is $2 kcal/g$. Is the claim of the engineerView Solution
- 9View SolutionAt constant pressure how much fraction of heat supplied to gas is converted into mechanical work?
- 10A container of volume $1{m^3}$is divided into two equal compartments by a partition. One of these compartments contains an ideal gas at $300 K$. The other compartment is vacuum. The whole system is thermally isolated from its surroundings. The partition is removed and the gas expands to occupy the whole volume of the container. Its temperature now would be ..... $K$View Solution