The rate of loss of heat from a body cooling under conditions of forced convection is proportional to its $(A)$ heat capacity $(B)$ surface area $(C)$ absolute temperature $(D)$ excess of temperature over that of surrounding : state if
Easy
(c)In forced convection rate of loss of heat $\frac{Q}{t} \propto A(T - {T_0})$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The coefficient of thermal conductivity of copper is nine times that of steel. In the composite cylindrical bar shown in the figure. What will be the temperature at the junction of copper and steel ....... $^oC$View Solution
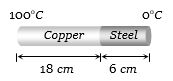
- 2The maximum energy in the thermal radiation from a hot source occurs at a wavelength of $11 \times {10^{ - 5}}cm$. According to Wein's law, the temperature of the source (on Kelvin scale) will be $n$ times the temperature of another source (on Kelvin scale) for which the wavelength at maximum energy is $5.5 \times {10^{ - 5}}cm$. The value $n$ isView Solution
- 3A body cools in $7$ minutes from $60^{\circ}\,C$ to $40^{\circ}\,C$. The temperature of the surrounding is $10^{\circ}\,C$. The temperature of the body after the next $7$ minutes will beView Solution
- 4If the ratio of coefficient of thermal conductivity of silver and copper is $10 : 9$ , then the ratio of the lengths upto which wax will melt in Ingen Hausz experiment will beView Solution
- 5A cup of tea cools from ${80^0}C$ to ${60^o}C$ in one minute. The ambient temperature is ${30^o}C$. In cooling from ${60^o}C$ to ${50^o}C$ it will take ....... $\sec$View Solution
- 6View SolutionIf a liquid is heated in weightlessness, the heat is transmitted through
- 7The temperature of a piece of iron is ${27^o}C$ and it is radiating energy at the rate of $Q\;kW{m^{ - 2}}$. If its temperature is raised to ${151^o}C$, the rate of radiation of energy will become approximately ....... $Q\,kW\,{m^{ - 2}}$View Solution
- 8View SolutionThe Wien’s displacement law express relation between
- 9Two thermometers $A$ and $B$ are exposed in sun light. The valve of $A$ is painted black, But that of $B$ is not painted. The correct statement regarding this case isView Solution
- 10A container with $1 kg$ of water in it is kept in sunlight, which causes the water to get warmer than the surroundings. The average energy per unit time per unit area received due to the sunlight is $700 Wm ^{-2}$ and it is absorbed by the water over an effective area of $0.05 m ^2$. Assuming that the heat loss from the water to the surroundings is governed by Newton's law of cooling, the difference (in ${ }^{\circ} C$ ) in the temperature of water and the surroundings after a long time will be. . . . . . . . (Ignore effect of the container, and take constant for Newton's law of cooling $=0.001 s ^{-1}$, Heat capacity of water $\left.=4200 J kg ^{-1} K ^{-1}\right)$View Solution
