The root mean square speed of smoke particles of mass $5 \times 10^{-17}\,kg$ in their Brownian motion in air at NTP is approximately $.......\,mm\,s ^{-1}$ [Given $k =1.38 \times 10^{-23}\,J\,K ^{-1}$ ]
JEE MAIN 2022, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1At $0 \;K$ which of the following properties of a gas will be zeroView Solution
- 2The rms speed of oxygen molecule in a vessel at particular temperature is $\left(1+\frac{5}{x}\right)^{\frac{1}{2}} v$, where $v$ is the average speed of the molecule. The value of $x$ will be:(Take $\pi=\frac{22}{7}$ )View Solution
- 3View SolutionThe quantity of heat required to raise one mole through one degree Kelvin for a monoatomic gas at constant volume is
- 4View SolutionMean kinetic energy (or average energy) per gm molecule of a monoatomic gas is given by
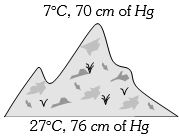
- 5At the top of a mountain a thermometer reads $7°C$ and a barometer reads $70\, cm$ of $Hg.$ At the bottom of the mountain these read $27°C$ and $76 \,cm$ of $Hg$ respectively. Comparison of density of air at the top with that of bottom isView Solution
- 6The $r.m.s.$ velocity will be greater forView Solution
- 7A gas has volume $V$ and pressure $P$. The total translational kinetic energy of all the molecules of the gas isView Solution
- 8A diatomic gas of molecular mass $40 \,g / mol$ is filled in a rigid container at temperature $30^{\circ} C$. It is moving with velocity $200 \,m / s$. If it is suddenly stopped, the rise in the temperature of the gas is .........View Solution
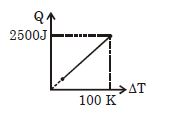
- 9One mole of a gas mixture is heated under constant pressure, and heat supplied $Q$ is plotted against temperature difference acquired. Find the approximate value of $\gamma $ for mixtureView Solution
- 10The temperature of argon, kept in a vessel, is raised by $1^\circ C$ at a constant volume. The total heat supplied to the gas is a combination of translational and rotational energies. Their respective shares areView Solution
