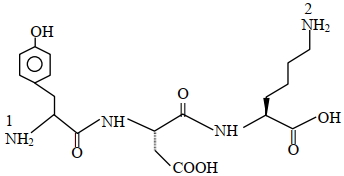
The structure of a peptide is given below
If the absolute values of the net charge of the peptide at $pH =2, pH =6$, and $pH =11$ are $\left| z _1\right|,\left| z _2\right|$ and $\left|z_3\right|$, respectively, then what is $\left|z_1\right|+\left|z_2\right|+\left|z_3\right|$ ?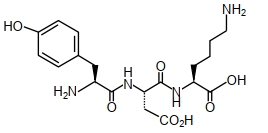
IIT 2020, Difficult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Molecular formula ${C_6}{H_{12}}{O_6}$ is ofView Solution
- 2The specific rotation of equilibrium mixture of $\alpha - D-$ glucose and $\beta - D-$ glucose, is ....... $^o$View Solution
- 3View SolutionIn fructose, the possible optical isomers are
- 4View SolutionAssertion :Disruption of the natural structure of a protein is called denaturation.
Reason : The change in colour and appearance of egg during cooking is due to denaturation - 5View SolutionIn the viscose process the solvent for cellulose consists of
- 6View SolutionThe helical structure of protein is stabilised by
- 7View SolutionGlucose in blood can be quantitatively determined with
- 8View SolutionThe vitamin which is water soluble and antioxidant is
- 9View SolutionWhich of the following does not show mutarotation
- 10A compound of mol. wt. $180$ is acetylated to give a compound of mol. wt. $390$. The number of amino groups in the initial compound isView Solution