The temperature at which the root mean square velocity of a molecule will be doubled than at $100°C$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1For one gram mol of a gas, the value of $R$ in the equation $PV = RT$ is nearly ...... $cal/K$View Solution
- 2View SolutionAccording to the kinetic theory of gases, total energy of a gas is equal to
- 3Consider a mixture of gas molecule of types $A, B$ and $C$ having masses $m_{A}\,<\,m_{B}\,<\,m_{C}$ ratio of their root mean square speeds at normal temperature and pressure is :View Solution
- 4The volume of a gas at $20°C$ is $200\, ml$. If the temperature is reduced to $-20°C$ at constant pressure, its volume will be ...... $ml$View Solution
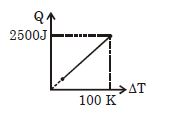
- 5One mole of a gas mixture is heated under constant pressure, and heat supplied $Q$ is plotted against temperature difference acquired. Find the approximate value of $\gamma $ for mixtureView Solution
- 6This question has Statement $- 1$ and Statement $-2$. Of the four choices given after the Statements, choose the one that best describes the two Statements.View Solution
Statement $1$ : The internal energy of a perfect gas is entirely kinetic and depends only on absolute temperature of the gas and not on its pressure or volume.
Statement $2$ : A perfect gas is heated keeping pressure constant and later at constant volume. For the same amount of heat the temperature of the gas at constant pressure is lower than that at constant volume. - 7View SolutionThe quantity of heat required to raise one mole through one degree Kelvin for a monoatomic gas at constant volume is
- 8View SolutionA piston is slowly pushed into a metal cylinder containing an ideal gas. Which of the following statements is/are incorrect?
- 9The molecular weight of a gas is $44$. The volume occupied by $2.2\, g$ of this gas at $0^\circ C$ and $2\, atm.$ pressure will be ....... $lit$View Solution
- 10A cylinder of fixed capacity of $44.8 \,litres$ contains helium gas at standard temperature and pressure. The amount of heat needed to raise the temperature of gas in the cylinder by $20.0^{\circ} C$ will be .............. $J$(Given gas constant $R =8.3 \,JK ^{-1}- moI ^{-1}$ )View Solution
