A cylinder of fixed capacity of $44.8 \,litres$ contains helium gas at standard temperature and pressure. The amount of heat needed to raise the temperature of gas in the cylinder by $20.0^{\circ} C$ will be .............. $J$(Given gas constant $R =8.3 \,JK ^{-1}- moI ^{-1}$ )
JEE MAIN 2022, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1One mole of a monoatomic ideal gas is expanded by a process described by $p V^3=C$, where $C$ is a constant. The heat capacity of the gas during the process is given by ( $R$ is the gas constant)View Solution
- 2The relation between root mean square speed $\left( v _{ rms }\right)$ and most probable speed $\left( v _{ p }\right)$ for the molar mass $M$ of oxygen gas molecule at the temperature of $300\, K$ will beView Solution
- 3For the $P-V$ diagram given for an ideal gas, out of the following which one correctly represents the $T-P$ diagram ?View Solution
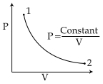
- 4The mean free path for a gas, with molecular diameter $d$ and number density $n$ can be expressed as :View Solution
- 5Two gases-argon (atomic radius $0.07 \;\mathrm{nm}$,atomic weight $40$ ) and xenon (atomic radius $0.1\; \mathrm{nm},$ atomic weight $140$ ) have the same number density and are at the same temperature. The raito of their respective mean free times is closest toView Solution
- 6The volume of a gas at pressure $21 \times {10^4}\,N/{m^2}$ and temperature $27^o C$ is $83 $ litres. If $R = 8.3\ J/mol/K$, then the quantity of gas in $gm-mole$ will beView Solution
- 7View SolutionA tyre kept outside in sunlight bursts off after sometime because of
- 8$5$ moles of oxygen is heated at constant volume from $10°C$ to $20°C.$ The change in the internal energy of the gas is (the gram molecular specific heat of oxygen at constant pressure...... $cal$ ${C_p} = 8$ $cal/mole \,°C$ and $R = 8.3 cal/mole\, °C)$View Solution
- 9The ratio of specific heats of a gas is $\frac{9}{7}$, then the number of degrees of freedom of the gas molecules for translational motion is :View Solution
- 10Let $\bar v , \bar v_{rms}$ and $v_p$ respectively denote the mean speed, root mean square speed and most probable speed of the molecules in an ideal monoatomic gas at absolute temperature $T$. The mass of the molecule is $m$. ThenView Solution
