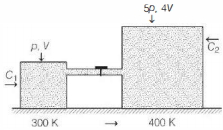
Two containers $C_{1}$ and $C_{2}$ of volumes $V$ and $4 \,V$ respectively, hold the same ideal gas and are connected by a thin horizontal tube of negligible volume with a valve which is initially closed. The initial pressures of the gas in $C_{1}$ and $C_{2}$ are $p$ and $5 p$, respectively. Heat baths are employed to maintain the temperatures in the containers at $300 \,K$ and $400 \,K$, respectively. The valve is now opened. Select the correct statement.
KVPY 2019, Advanced
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1At constant temperature on increasing the pressure of a gas by $5\%$ will decrease its volume by ..... $\%$View Solution
- 2Air is filled in a bottle at atmospheric pressure and it is corked at $35°C.$ If the cork can come out at $3$ atmospheric pressure than upto what temperature should the bottle be heated in order to remove the cork ...... $^oC$View Solution
- 3View SolutionOn colliding in a closed container the gas molecules
- 4An insulated box containing a diatomic gas of molar mass $M$ is moving with a velocity $v$. The box is suddenly stopped. The resulting change in temperature isView Solution
- 5A jar contains a gas and few drops of water at $T K.$ The pressure in the jar is $830 \,mm$ of mercury. The temperature of jar is reduced by $1\%.$ The saturated vapour pressure of water at the two temperatures are $30 \,mm$ and $ 25\, mm$ of mercury. Then the new pressure in the jar will be ..... $mm$ of $Hg$View Solution
- 6View SolutionThe pressure exerted by the gas on the walls of the container because
- 7The plot that depicts the behavior of the mean free time $t$ (time between two successive collisions) for the molecules of an ideal gas, as a function of temperature $(T)$, qualitatively, is (Graphs are schematic and not drawn to scale)View Solution
- 8View SolutionSupposing the distance between the atoms of a diatomic gas to be constant, its specific heat at constant volume per mole (gram mole) is
- 9$12\,\,gms$ of gas occupy a volume of $4\times 10^{-3}\,\, m^3$ ata temperature of $7\,^oC$. After the gas is heated at constant pressure its density becomes $6\times 10^{-4}\,\,gm/cc$. ...... $K$ temperature to which the gas was heated.View Solution
- 10View SolutionIf the pressure in a closed vessel is reduced by drawing out some gas, the mean free path of the molecules