A jar contains a gas and few drops of water at $T K.$ The pressure in the jar is $830 \,mm$ of mercury. The temperature of jar is reduced by $1\%.$ The saturated vapour pressure of water at the two temperatures are $30 \,mm$ and $ 25\, mm$ of mercury. Then the new pressure in the jar will be ..... $mm$ of $Hg$
Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
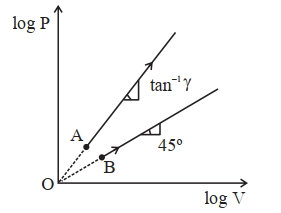
- 1Two thermodynamical process are shown in the figure. The molar heat capacity for process $A$ and $B$ are $C_A$ and $C_B$. The molar heat capacity at constant pressure and constant volume are represented by $C_P$ and $C_V$, respectively. Choose the correct statement.View Solution
- 2View SolutionIf a Vander-Waal's gas expands freely, then final temperature is
- 3View SolutionIf two moles of diatomic gas and one mole of mono atomic gas are mixed then the ratio of specific heats is
- 4A gaseous mixture consists of $16\,g$ of helium and $16\,g$ of oxygen. The ratio $\frac{{{C_P}}}{{{C_V}}}$ of the mixture isView Solution
- 5An open and wide glass tube is immersed vertically in mercury in such a way that length $0.05\,\, m$ extends above mercury level. The open end of the tube is closed and the tube is raised further by $0.43 \,\,m$. The length of air column above mercury level in the tube will be ...... $m$ Take $P_{atm} = 76 \,\,cm$ of mercuryView Solution
- 6Statement$-1 :$ Internal energy of gas $U = nC_VT$ is due to random motion of gas molecules.View Solution
Statement$-2 :$ A container is moving with speed $v$. It is suddenly stopped by a force, temperature of gas increases. - 7For a gas, the $r.m.s.$ speed at $800\, K$ isView Solution
- 8One mole of an ideal gas requires $207\, J$ heat to raise the temperature by $10 \,K$ when heated at constant pressure. If the same gas is heated at constant volume to raise the temperature by the same $10\, K,$ the heat required is ...... $J$View Solution
(Given the gas constant $R = 8.3J/mol{\rm{ - }}K$)
- 9Two bulbs of identical volumes connected by a small capillary are initially filled with an ideal gas at temperature $T$. Bulb $2$ is heated to maintain a temperature $2 T$, while bulb $1$ remains at temperature $T$. Assume throughout that the heat conduction by the capillary is negligible. Then, the ratio of final mass of the gas in bulb $2$ to the initial mass of the gas in the same bulb is close toView Solution
- 10Two gases occupy two containers $A$ and $B$ the gas in $A$, of volume $0.10\,m ^3$, exerts a pressure of $1.40\,MPa$ and that in $B$ of volume $0.15 m ^3$ exerts a pressure $0.7\,MPa$. The two containers are united by a tube of negligible volume and the gases are allowed to intermingle. Then if the temperature remains constant, the final pressure in the container will be (in MPa)View Solution
