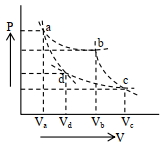
$\mathrm{TV}^{\gamma-1}=\text { constant }$
$\mathrm{T}_{\mathrm{a}} \cdot \mathrm{V}_{\mathrm{a}}^{\gamma-1}=\mathrm{T}_{\mathrm{d}} \cdot \mathrm{V}_{\mathrm{d}}^{\gamma-1}$
$\left(\frac{\mathrm{V}_{\mathrm{a}}}{\mathrm{V}_{\mathrm{d}}}\right)^{\gamma-1}=\frac{\mathrm{T}_{\mathrm{d}}}{\mathrm{T}_{\mathrm{a}}}$
$\mathrm{T}_{\mathrm{b}} \cdot \mathrm{V}_{\mathrm{b}}^{\gamma-1}=\mathrm{T}_{\mathrm{c}} \cdot \mathrm{V}_{\mathrm{c}}^{\gamma-1}$
$\left(\frac{\mathrm{V}_{\mathrm{b}}}{\mathrm{V}_{\mathrm{c}}}\right)^{\gamma-1}=\frac{\mathrm{T}_{\mathrm{c}}}{\mathrm{T}_{\mathrm{b}}}$
$\mathrm{T}_{\mathrm{b}} \cdot \mathrm{V}_{\mathrm{b}}^{\gamma-1}=\mathrm{T}_{\mathrm{c}} \cdot \mathrm{V}_{\mathrm{c}}^{\gamma-1}$
$\left(\frac{\mathrm{V}_{\mathrm{b}}}{\mathrm{V}_{\mathrm{c}}}\right)^{\gamma-1}=\frac{\mathrm{T}_{\mathrm{c}}}{\mathrm{T}_{\mathrm{b}}}$
$\frac{\mathrm{V}_{\mathrm{a}}}{\mathrm{V}_{\mathrm{d}}}=\frac{\mathrm{V}_{\mathrm{b}}}{\mathrm{V}_{\mathrm{c}}} \quad\left(\begin{array}{r}\because \mathrm{T}_{\mathrm{d}}=\mathrm{T}_{\mathrm{c}} \\ \mathrm{T}_{\mathrm{a}}=\mathrm{T}_{\mathrm{b}}\end{array}\right)$
Download our appand get started for free
Similar Questions
- 1On a $TP$ diagram, two moles of ideal gas perform process $AB$ and $CD$. If the work done by the gas in the process $AB$ is two times the work done in the process $CD$ then what is the value of $T_1/T_2$?View Solution
- 2Initial pressure and volume of a gas are $P$ and $V$ respectively. First it is expanded isothermally to volume $4V$ and then compressed adiabatically to volume $V$ . The final pressure of gas will be (given $\gamma = 3/2$ )View Solution
- 3View SolutionOut of the following which quantity does not depend on path
- 4View SolutionWhich is incorrect
- 5Consider the efficiency of Carnot's engine is given by $\eta=\frac{\alpha \beta}{\sin \theta} \log _{e} \frac{\beta x}{k T}$, where $\alpha$ and $\beta$ are constants. If $T$ is temperature, $k$ is Boltzman constant, $\theta$ is angular displacement and $x$ has the dimensions of length. Then, choose the incorrect option.View Solution
- 6An ideal gas is expanding such that $\mathrm{PT}^2=$ constant. The coefficient of volume expansion of the gas isView Solution
- 7View SolutionIn a thermodynamic system working substance is ideal gas, its internal energy is in the form of
- 8View SolutionWhen heat is given to a gas in an isothermal change, the result will be
- 9An ideal gas expands isothermally from a volume ${V_1}$ to ${V_2}$ and then compressed to original volume ${V_1}$adiabatically. Initial pressure is ${P_1}$ and final pressure is ${P_3}$. The total work done is $W$. ThenView Solution
- 10One mole of an ideal gas is taken through an adiabatic process where the temperature rises from $27^{\circ} {C}$ to $37^{\circ} {C}$. If the ideal gas is composed of polyatomic molecule that has $4$ vibrational modes which of the following is true?View Solution
