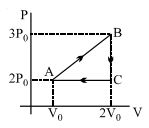
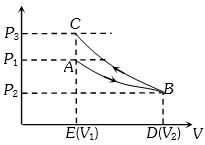
An ideal gas expands isothermally from a volume ${V_1}$ to ${V_2}$ and then compressed to original volume ${V_1}$adiabatically. Initial pressure is ${P_1}$ and final pressure is ${P_3}$. The total work done is $W$. Then
IIT 2004, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$A$ reversible engine converts one-sixth of the heat input into work. When the temperature of the sink is reduced by $ 62^oC$, the efficiency of the engine is doubled. The temperatures of the source and sink areView Solution
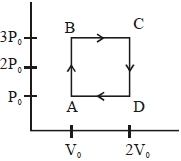
- 2An engine operates by taking a monatomic ideal gas through the cycle shown in the figure. The percentage efficiency of the engine is close to $.......\%$View Solution
- 3Determine coefficient of performance of given temperature limit.View Solution
$T_{1}=27^{\circ} C$ [outside fridge]
$T_{2}=-23^{\circ} C$ [inside fridge]
- 4An ideal gas at atmospheric pressure is adiabatically compressed so that its density becomes $32$ times of its initial value. If the final pressure of gas is $128$ atmosphers, the value of $\gamma$ the gas isView Solution
- 5In a Carnot engine, when ${T_2} = {0^o}C$ and ${T_1} = {200^o}C,$ its efficiency is ${\eta _1}$ and when ${T_1} = 0{\,^o}C$ and ${T_2} = - 200{\,^o}C$, Its efficiency is ${\eta _2}$, then what is ${\eta _1}/{\eta _2}$View Solution
- 6View SolutionIn the case of diatomic gas, the heat given at constant pressure is that part of energy which is used for the expansion of gas, is
- 7A Carnot engine operates between two reservoirs of temperatures $900\; \mathrm{K}$ and $300 \;\mathrm{K}$ The engine performs $1200\; \mathrm{J}$ of work per cycle. The heat energy (in $\mathrm{J}$ ) delivered by the engine to the low temperature reservoir, in a cycle. isView Solution
- 8A Carnot engine take $5000 \,k\,cal$ of heat from a reservoir at $727\,^{\circ}C$ and gives heat to a $\operatorname{sink}$ at $127\,^{\circ}C$. The work done by the engine is $.......... \times 10^{6}\,J$View Solution
- 9Consider the efficiency of Carnot's engine is given by $\eta=\frac{\alpha \beta}{\sin \theta} \log _{e} \frac{\beta x}{k T}$, where $\alpha$ and $\beta$ are constants. If $T$ is temperature, $k$ is Boltzman constant, $\theta$ is angular displacement and $x$ has the dimensions of length. Then, choose the incorrect option.View Solution
- 10View SolutionIn the above thermodynamic process, the correct statement is