Two substances of densities ${\rho _1}$ and ${\rho _2}$ are mixed in equal volume and the relative density of mixture is $4$. When they are mixed in equal masses, the relative density of the mixture is $3$. The values of ${\rho _1}$ and ${\rho _2}$ are
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionPressure applied to an enclosed fluid is transmitted undiminished to every portion of the fluid and the walls of the containing vessel. This law was first formulated by
- 2View SolutionThe displacement of a ball falling from rest in a viscous medium is platted against time. Choose a possible option
- 3Two bodies are in equilibrium when suspended in water from the arms of a balance. The mass of one body is $36 g $ and its density is $9 g / cm^3$. If the mass of the other is $48 g$, its density in $g / cm^3$ isView Solution
- 4The flow speeds of air on the lower and upper surfaces of the wing of an aeroplane are $v$ and $2v$ respectively. The density of air is $\rho $ and surface area of wing is $A$ . The dynamic lift on the wing isView Solution
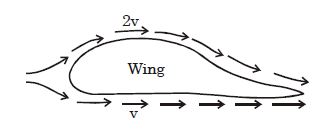
- 5A block of steel of size $ 5 cm × 5 cm × 5 cm $ is weighed in water. If the relative density of steel is $7,$ its apparent weight isView Solution
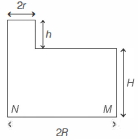
- 6A cylindrical vessel of base radius $R$ and height $H$ has a narrow neck of height $h$ and radius $r$ at one end (see figure). The vessel is filled with water (density $\rho_w$ ) and its neck is filled with immiscible oil (density $\rho_0$ ). Then, the pressure atView Solution
- 7There is a hole of area $A$ at the bottom of cylindrical vessel. Water is filled up to a height $ h$ and water flows out in $ t $ second. If water is filled to a height $4h,$ it will flow out in time equal toView Solution
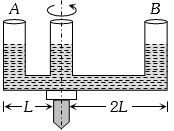
- 8A given shaped glass tube having uniform cross section is filled with water and is mounted on a rotatable shaft as shown in figure. If the tube is rotated with a constant angular velocity $\omega $thenView Solution
- 9View SolutionViscosity is the property of a liquid due to which it :
- 10The velocity of upper layer of water in a river is $36 kmh ^{-1}$. Shearing stress between horizontal layers of water is $10^{-3} Nm ^{-2}$. Depth of the river is $m$. (Co-efficiency of viscosity of water is $10^{-2} \,Pa . s$ )View Solution
