When ethanoic acid reacts with sodium hydrogencarbonate, then a salt X is formed and a gas Y is evolved.
Name the salt X and gas Y. Describe an activity with the help of a labelled diagram of the apparatus used to prove that the evolved gas is the one which you have named. Also write the chemical equation of the reaction involved.
Name the salt X and gas Y. Describe an activity with the help of a labelled diagram of the apparatus used to prove that the evolved gas is the one which you have named. Also write the chemical equation of the reaction involved.
Salt X is sodium ethanoate, $CH _3 COONa$; Gas Y is carbon dioxide, $CO _2$.
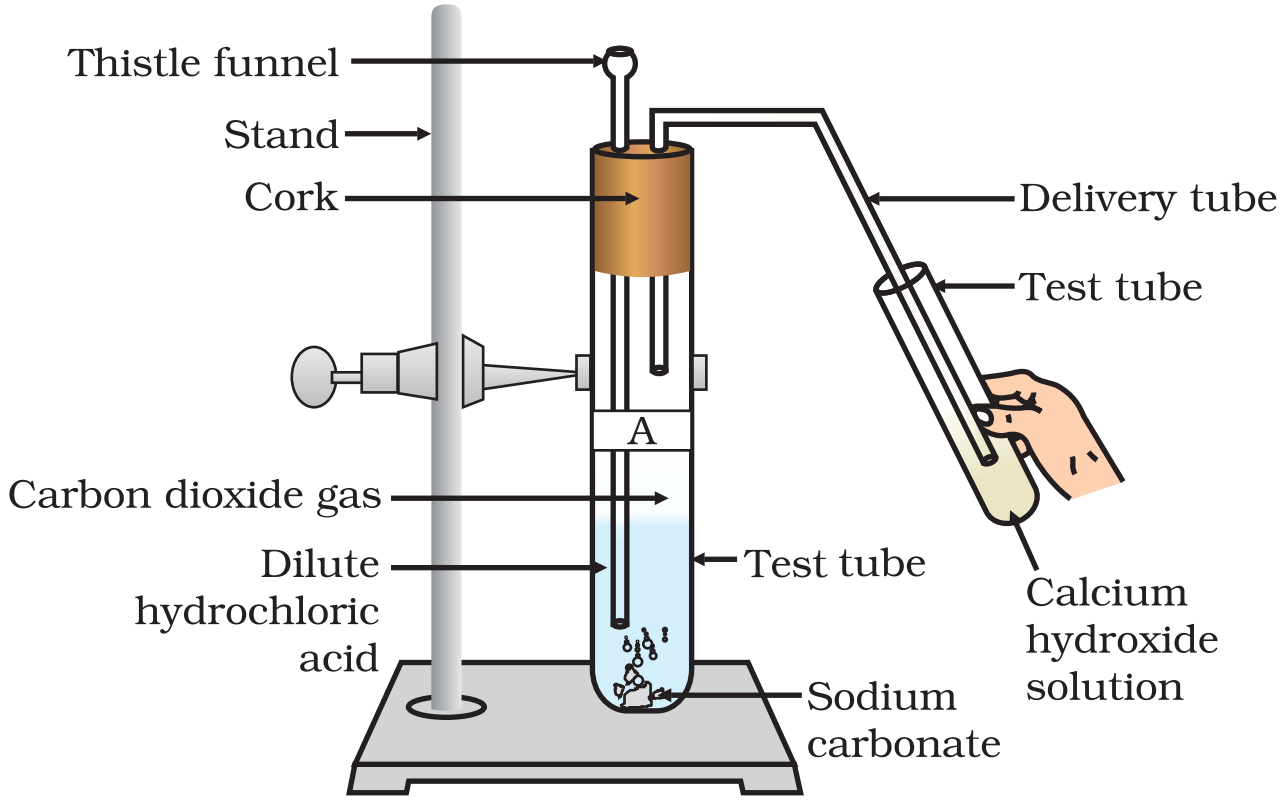
Activity: Take a boiling tube and put about 0.5g of sodium carbonate in it. Add 2ml of dilute ethanoic acid to the boiling tube (through a thistle funnel). We will observe that brisk effervescence of carbon dioxide gas is produced. Let us pass this gas through lime water taken in a test tube. We will find that lime water turns milky. Only carbon dioxide gas can turn lime water milky. So, this experiment proves that when ethanoic acid reacts with sodium carbonate, then carbon dioxide gas is evolved.
$\text{CH}_3\text{COOH}+\text{NaHCO}_3\rightarrow\text{CH}_3\text{COONa}+\text{CO}_2\uparrow+\text{H}_2\text{O} $
Activity: Take a boiling tube and put about 0.5g of sodium carbonate in it. Add 2ml of dilute ethanoic acid to the boiling tube (through a thistle funnel). We will observe that brisk effervescence of carbon dioxide gas is produced. Let us pass this gas through lime water taken in a test tube. We will find that lime water turns milky. Only carbon dioxide gas can turn lime water milky. So, this experiment proves that when ethanoic acid reacts with sodium carbonate, then carbon dioxide gas is evolved.

$\text{CH}_3\text{COOH}+\text{NaHCO}_3\rightarrow\text{CH}_3\text{COONa}+\text{CO}_2\uparrow+\text{H}_2\text{O} $
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Consider the following organic compounds:View Solution
$CH _3 OH , C _2 H _5 OH , CH _3 COCH _3, CH _3 COOH , C _2 H _5 COOH , C _4 H _9 COOC _2 H _5, CH _4, C _2 H _6, CH _3 CHO , HCHO$
Out of these compounds:
a. Which compound is most likely to be sweet-smelling?
b. Which compound on treatment with conc. $H _2 SO _4$ at $170^{\circ} C$ forms an alkene?
c. Which compound on repeated chlorination forms chloroform?
d. Which compound is added to alcohol to denature it?
e. Which compound is a constituent of vinegar?
f. Which compound is used to sterilise wounds and syringes? - 2View SolutionDescribe the structure of graphite with the help of a labelled diagram.
- 3A colourless organic liquid $X$ of molecular formula $C _2 H _4 O _2$ turns blue litmus to red. Another colourless organic liquid Y of molecular formula $C _3 H _6 O$ has no action on any litmus but it is used as a nail polish remover. A yet another colourless organic liquid Z of molecular formula $C _2 H _6 O$ has also no action on litmus but it is used in tincture of iodine.View Solution
a. Name the liquid X . To which homologous series does it belong? Give the name of another member of this homologous series.
b. Name the liquid Y. To which homologous series does it belong? Write the name of another member of this homologous series.
c. Can you name an organic compound having the same molecular formula as liquid $Y$ but which belongs to a different homologous series? What is this homologous series?
d. Name the liquid Z. To which homologous series does it belong? Write the name of another member of this homologous series. - 4An organic compound A (molecular formula $C _2 H _4 O _2$ ) reacts with Na metal to form a compound B and evolves a gas which burns with a pop sound. Compound A on treatment with an alcohol C in the presence of a little of concentrated sulphuric acid forms a sweet-smelling compound D (molecular formula $C _3 H _6 O _2$ ). Compound D on treatment with NaOH solution gives back B and C. Identify A, B, C and D.View Solution
- 5Two organic compounds A and B have the same molecular formula $C_6H_{12}$. Write the names and structural formulae:View Solution
- If A is a cyclic compound.
- If B is an open chain compound.
- Which compound contains single bonds as well as a double bond?
- Which compound contains only single bonds?
- 6A four carbons atoms containing neutral organic compound $X$ reacts with sodium metal to evolve a gas which burns with 'pop' sound. Another four carbon atoms containing carbon compound reacts with sodium hydrogen carbonate to evolve a gas which turns lime water milky. When compounds $X$ and $Y$ are heated together in the presence of a little of concentrated sulphuric acid, then a new compound $Z$ is formed.View Solution
a. What is compound $X$ ? Also write its formula.
b. What is compound Y? Also write its formula.
c. What is compound Z? Also write its formula.
d. What type of smell is given by compound Z?
e. What is the general name of compounds like $Z$ ?
f. What is the general name of the reaction which which takes place between $X$ and $Y$ to form $Z$ ? - 7View SolutionExplain the cleansing action of soap. Draw diagrams to illustrate your answer.
- 8An organic compound $A$ having the molecular formula $C _3 H _8 O$ is a liquid at room temperature. The organic liquid $A$ reacts with sodium metal to evolve a gas which burns causing a little explosion. When the organic liquid A heated with concentrated sulphuric acid at $170^{\circ} C$, it forms a compound B which decolourizes bromine water. The compound B adds on one molecule of hydrogen in the presence of Ni as catalyst to forms compound C which gives substitution reactions with chlorine.View Solution
a. What is compound A ?
b. What is compound B ?
c. What type of reaction occurs when A is converted into B ?
d. What is compound C?
e. What type of reaction takes place when $B$ is converted into $C$ ? - 9View SolutionDescribe the structure of diamond. Draw a simple diagram to show the arrangement of carbon atoms in diamond.
- 10An organic compound having the molecular formula $C_3H_6O$ can exist in the form of two isomers A and B having different functional groups. The isomer A is a liquid which is used as a solvent for nail polish. The isomer B is also a liquid. An aqueous solution of one of the lower homologues of B is used for preserving biological specimens in the laboratory.View Solution
- What is compound A?
- Write the electron-dot structure of A.
- What is compound B?
- Write the electron-dot structure of B.
- Name the lower homologue of compound B which is used in preserving biological specimens.
