Which of the following compounds is aromatic alcohol?
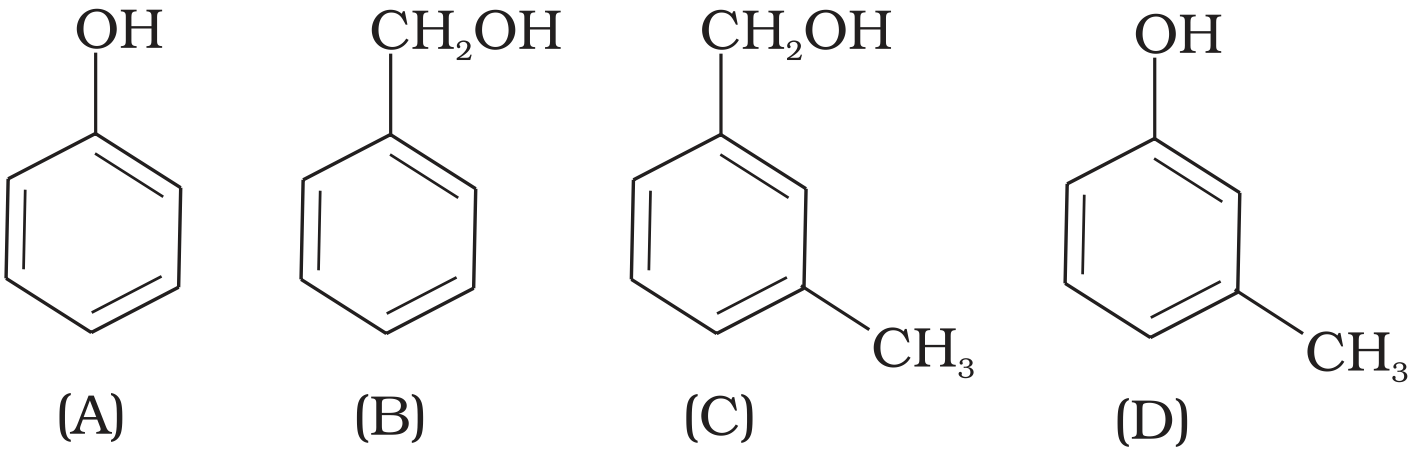

Compound $(A)$ i.e., phenol and compound $(D)$ i.e., a derivative of phenol cannot be considered as aromatic alcohol. As phenol is also known as, carbolic acid cannot be considered as aromatic alcohol.
Compound $(B)$ and $(C), -OH$ group is bonded to $sp^3$ hybridised carbon which in turn is bonded to benzene ring.
Compound $(B)$ and $(C), -OH$ group is bonded to $sp^3$ hybridised carbon which in turn is bonded to benzene ring.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionWhat is the hybridisation of carbon and oxygen in electronic structure of ether?
- 2Give IUPAC name of the compound given below.View Solution
$\text{CH}_3-\text{CH}-\text{CH}_2-\text{CH}_2-\text{CH}-\text{CH}_3 \\ \ \ \ \ \ \ \ \ \ \ \ \ \ \ | \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ | \\ \ \ \ \ \ \ \ \ \ \ \ \ \ \text{Cl} \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \text{CH}$ - 3View SolutionThe major product obtained on acid - catalysed hydration of 2-phenylpropene is:
- 4View SolutionThe suffix used in the nomenclature of alcohol is:
- 5Write the $\ce{IUPAC}$ name of the given compound:$\text{HO}−\text{CH}_2−\text{CH}−\text{CH}_2−\text{OH}\\ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ ∣ \\ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \text{CH}_3$View Solution
- 6View SolutionThe order of boiling point of primary (1), secondary (2) and tertiary (3) alcohols is:
- 7View SolutionTrivial name of methanol is _________.
- 8View SolutionMark the correct statement:
- 9Carbinol is a trivial name of$:$View Solution
- 10View SolutionWhich of the following combinations can be used for the preparation of cis vic diol?
