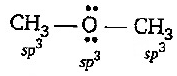Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionWhich reagent is suitable for this conversion?
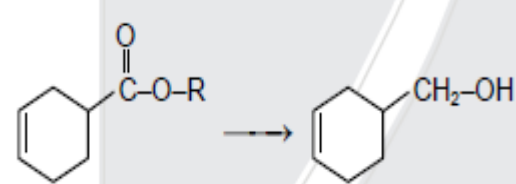
- 2View SolutionMark the correct order of decreasing acid strength of the following compounds.
- 3The best method to prepare $3-$methylbutan$-2-ol$ from $3-$methylbut$-1-$ene is:View Solution
- 4View SolutionWhich of the following alcohols is the least soluble in water?
- 5View SolutionMixed ether will not be formed in the reaction?
- 6View SolutionAlcohols containing only up to ________________ carbon atoms are completely miscible with water.
- 7View SolutionThe IUPAC name is:
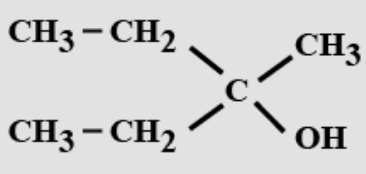
- 8View SolutionThe IUPAC name of the following compound is:
- 9View SolutionEthanol on reaction with acetic anhydride gives:
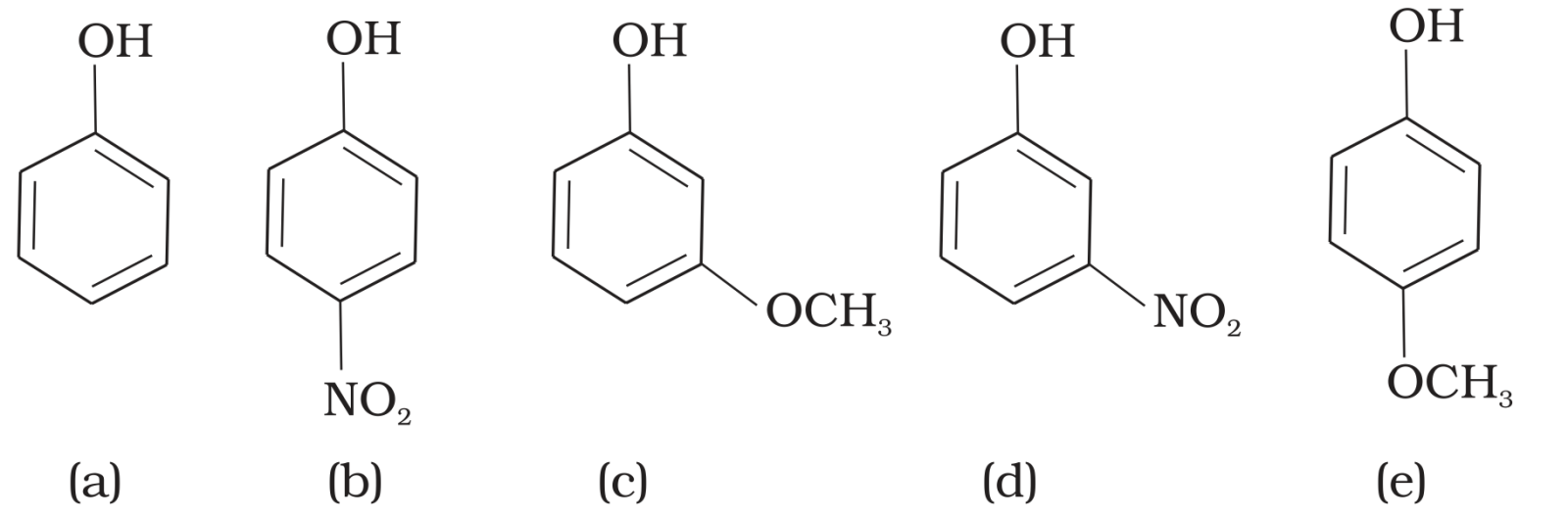
- 10View SolutionThe strongest acid in the following compounds is: