With the help of a labelled diagram, describe the method of separating ammonium chloride from a mixture of ammonium chloride and common salt. Mention the difference in the properties of ammonium chloride and sodium chloride which has made this separations possible.
The mixture of common salt and ammonium chloride is taken in a china dish and placed on a tripod stand. The china dish is covered with an inverted glass funnel. A loose cotton plug is put in the upper, open end of the funnel to prevent the ammonium chloride vapours from escaping into the atmosphere. The china dish is heated by using a burner. On heating the mixture, ammonium chloride changes into white vapours. These vapours rise up and get converted into solid ammonium chloride on coming in contact with the cold, inner walls of the funnel. In this way, pure ammonium chloride collects on the inner sides of the funnel in the form of a sublimate and can be removed. Common salt does not change into vapours on heating, so it remains behind in the china dish and can be separated out. 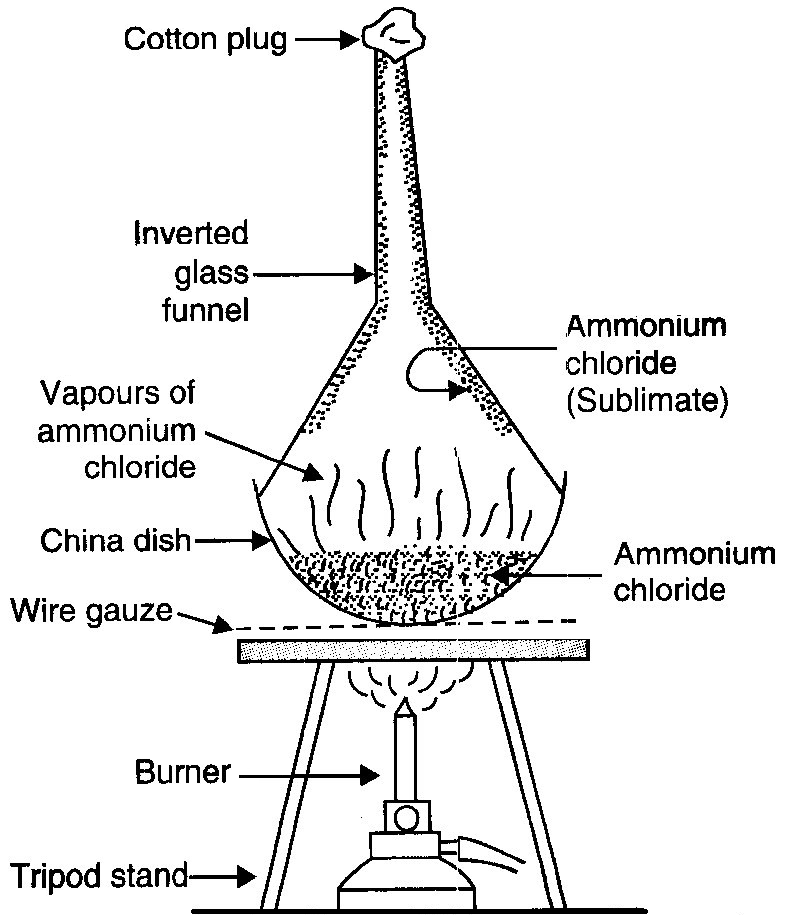 Ammonium chloride sublimes on heating whereas common salt does not sublime on heating. So, we can separate ammonium chloride from a mixture of common salt and ammonium chloride by the process of sublimation.
Ammonium chloride sublimes on heating whereas common salt does not sublime on heating. So, we can separate ammonium chloride from a mixture of common salt and ammonium chloride by the process of sublimation.
 Ammonium chloride sublimes on heating whereas common salt does not sublime on heating. So, we can separate ammonium chloride from a mixture of common salt and ammonium chloride by the process of sublimation.
Ammonium chloride sublimes on heating whereas common salt does not sublime on heating. So, we can separate ammonium chloride from a mixture of common salt and ammonium chloride by the process of sublimation.Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionDraw a labelled diagram of the fractional distillation apparatus used for separating a mixture of alcohol and water.
- 2View SolutionDiscuss the method of separating a mixture containing chalk powder, iron fillings and naphthalene.
- 3View SolutionDefine the following:
- Sol
- Aerosol
- Emulsion
- Foam
- 4View SolutionHow will you separate a mixture of mercury, oil and water?
- 5There are three substances $X, Y$ and $Z.$ The substance $X$ does not have a fixed melting point or boiling point and it still shows the individual properties of its constituents. The substance $Y$ is a pure substance which occurs in nature as such. The substance $Y$ has a fixed melting point and boiling point but it cannot be broken down into simpler substances by any chemical means. The substance $Z$ is also a pure substance whose properties are entirely different from those of its constituents. The substance $Z$ can, however, be divided by electrolysis into two substances which belong to the same class of substances as $Y.$View Solution
- What type of substance could $X$ be$?$ Name one substance like $X.$
- What type of substance could $Y$ be$?$ Name one substance like $Y.$
- What type of substance could $Z$ be$?$ Name one substance like $Z.$
- Whish process involves absorption or release of an appreciable amount of energy: formation of substance $X$ or formation of substance $Z?$
- Name the three groups into which all the substances like $Y$ are divided on the basis of their properties.
- 6View SolutionDifferentiate between a saturated and an unsaturated solution. How will you test whether a given solution is saturated or not?
- 7View SolutionHow will you separate camphor, common salt and iron nails from their mixture?
- 8View SolutionList five characteristics by which compounds can be distinguished from mixtures.
- 9How would you prepare a saturated solution of sodium chloride in water at $25^\circ C?$ What will happen if this solution is cooled to $10^\circ C?$View Solution
- 10View SolutionGive the main differences between physical changes and chemical changes.
