Work done by a Carnot engine operating between temperatures $127^{\circ}\,C$ and $27^{\circ}\,C$ is $2\,kJ$. The amount of heat transferred to the engine by the reservoir is $........\,kJ$
JEE MAIN 2023, Medium
$\eta=1-\frac{ T _2}{ T _1}=\frac{ W }{ Q _1}$
$\Rightarrow \frac{ W }{ Q _1}=1-\frac{300}{400}=\frac{1}{4}$
$\Rightarrow \frac{2 kJ }{ Q _1}=\frac{1}{4}$
$\Rightarrow Q _1=8\,kJ$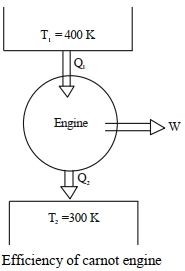
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The volume of a gas is reduced adiabatically to $\frac{1}{4}$ of its volume at $27°C$, if the value of $\gamma = 1.4,$ then the new temperature will beView Solution
- 2A certain amount of gas is taken through a cyclic process $(A\,B\,C\,D\,A)$ that has two isobars, one isochore and one isothermal. The cycle can be represented on a $P-V$ indicator diagram asView Solution
- 3View SolutionThe door of a working refrigerator is left open in a well insulated room. The temperature of air in the room will
- 4Initial pressure and volume of a gas are $P$ and $V$ respectively. First it is expanded isothermally to volume $4V$ and then compressed adiabatically to volume $V$ . The final pressure of gas will be (given $\gamma = 3/2$ )View Solution
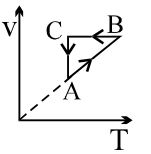
- 5View SolutionAn ideal gas undergoes a thermodynamics cycle as shown in figure. Which of the following graphs represents the same cycle?
- 6A monatomic gas at pressure $P_1$ and volume $V_1$ is compressed adiabatically to ${\frac{1}{8}}^{th}$ of its original volume. What is the final pressure of the gas is ........ $P_1$?View Solution
- 7The volume of an ideal gas $(\gamma=1.5)$ is changed adiabatically from $5$ litres to $4$ litres. The ratio of initial pressure to final pressure is:View Solution
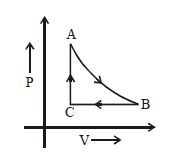
- 8The adjoining figure shows the $P-V$ diagram for a fixed mass of an ideal gas undergoing cyclic process. $AB$ represents isothermal process and $CA$ represents isochoric process.Which of the graph shown in the following figures represents the $P-T$ diagram of the cyclic process ?View Solution
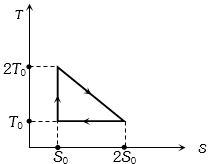
- 9View SolutionThe temperature-entropy diagram of a reversible engine cycle is given in the figure. Its efficiency is
- 10A Carnot's heat engine works between the temperatures $427^{\circ} C$ and $27^{\circ} C$. $...........\,kcal / s$ amount of heat should it consume per second to deliver mechanical work at the rate of $1.0\,kW$View Solution
