The efficiency of the heat engine is
$\eta=1-\frac{T_2}{T_1}=1-\left(\frac{273+27 K}{273+427 K}\right)=\frac{4}{7}$
But $\eta=\frac{ W }{ Q _1}$
$\therefore Q _1=\frac{ W }{\eta}=\frac{1.0 kW }{4 / 7}=1.75 kW =0.417 kcal / s$
Thus, the engine would require $417 cal$ of heat per second, to deliver the requisite amount of work.
Download our appand get started for free
Similar Questions
- 1A Carnot engine whose low temperature reservoir is at $7\,°C$ has an efficiency of $50\%$. It is desired to increase the efficiency to $70\%$. By how many degrees should the temperature of the high temperature reservoir be increased ....... $K$View Solution
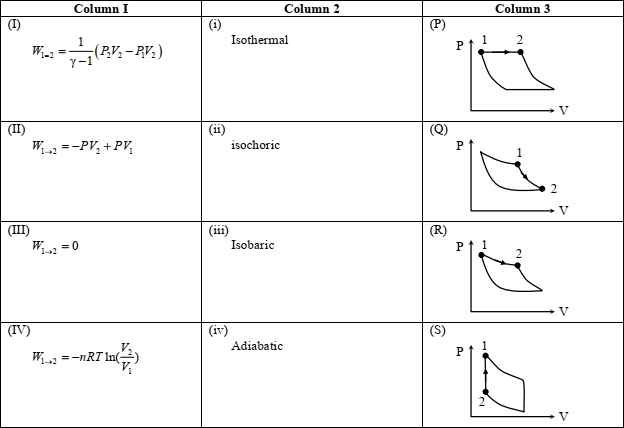
- 2An ideal gas is undergoing a cyclic thermodynamic process in different ways as shown in the corresponding $P$ $V$ diagrams in column $3$ of the table. Consider only the path from state $1$ to $2 . W$ denotes the corresponding work done on the system. The equations and plots in the table have standard notations as used in thermodynamic processes. Here $\gamma$ is the ratio of heat capacities at constant pressure and constant volume. The number of moles in the gas is $n$.View Solution
(image)
($1$) Which of the following options is the only correct representation of a process in which $\Delta U=\Delta Q-P \Delta V$ ?
$[A] (II) (iv) (R)$ $[B] (II) (iii) (P)$ $[C] (II) (iii) (S)$ $[D] (III) (iii) (P)$
($2$) Which one of the following options is the correct combination?
$[A] (III) (ii) (S)$ $[B] (II) (iv) (R)$ $[C] (II) (iv) (P)$ $[D] (IV) (ii) (S)$
($3$) Which one of the following options correctly represents a thermodynamic process that is used as a correction in the determination of the speed of sound in an ideal gas?
$[A] (III) (iv) (R)$ $[B] (I) (ii)$ $(\mathrm{Q})$ $[C] (IV) (ii) (R)$ $[D] (I) (iv) (Q)$
- 3View SolutionWhen heat is given to a gas in an isothermal change, the result will be
- 4An ideal gas expands from volume $V_1$ to $V_2$. This may be achieved by either of the three processes: isobaric, isothermal and adiabatic. Let $\Delta U$ be the change in internal energy of the gas, $Q$ be the quantity of heat added to the system and $W$ be the work done by the system on the gas. Identify which of the following statements is false for $\Delta U$?View Solution
- 5When an ideal gas $(\gamma = 5/3$) is heated under constant pressure, then what percentage of given heat energy will be utilised in doing external workView Solution
- 6$Assertion :$ In isothermal process whole of the heat supplied to the body is converted into internal energy.View Solution
$Reason :$ According to the first law of thermodynamics : $\Delta Q = \Delta U + p\Delta V$ - 7View SolutionIn which thermodynamic process, volume remains same
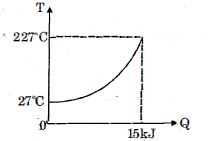
- 8$Q$ amount of heat is given to $0.5\ mole$ of an ide al mono-atomic gas by a process $TV^n$ constant. Following graph shows variation of temperature with $Q$ . Find value of $n$.View Solution
- 9Two identical containers $A$ and $B$ with frictionless pistons contain the same ideal gas at the same temperature and the same volume $V$. The mass of the gas in $A$ is ${m_A}$ and that in $B$ is ${m_B}$. The gas in each cylinder is now allowed to expand isothermally to the same final volume $2V$. The changes in the pressure in $A$ and $B$ are found to be $\Delta P$ and $1.5 \Delta P$ respectively. ThenView Solution
- 10For an ideal gas, the internal energy is given by $U=5 p V / 2+C$, where $C$ is a constant. The equation of the adiabats in the $p V$-plane will beView Solution
