$5.6$ $liter$ of helium gas at $STP$ is adiabatically compressed to $0.7$ $liter$. Taking the initial temperature to be $T_1$, the work done in the process is
Diffcult
No. of moles $=n=\frac{5.6}{22.4}=\frac{1}{4}$
$\mathrm{TV}^{\gamma -1}=$ constant $\Rightarrow \mathrm{T}_{1}(5.6)^{2 / 3}=\mathrm{T}_{2}(0.7)^{2 / 3}$
$\Rightarrow \mathrm{T}_{1}(8)^{2 / 3}=\mathrm{T}_{2} \Rightarrow 4 \mathrm{T}_{1}=\mathrm{T}_{2}$
$\mathrm{W}_{\mathrm{gas}}=\frac{-n R \Delta T}{\gamma-1}=-\frac{(1) R\left(3 T_{1}\right) \times 3}{4 \times 2}=-\frac{9}{8} R T_{1}$
Therefore $\mathrm{W}_{\text {external}}=\frac{9}{8} R T_{1}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$Assertion :$ When a bottle of cold carbonated drink is opened, a slight fog forms around the opening.View Solution
$Reason :$ Adiabatic expansion of the gas causes lowering of temperature and condensation of water vapours. - 2If $\gamma = 2.5$ and volume is equal to $\frac{1}{8}$ times to the initial volume then pressure $P' $ is equal to (Initial pressure $= P$)View Solution
- 3View SolutionWhich of the following processes is reversible
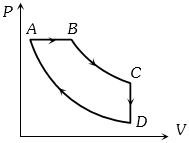
- 4A cyclic process $ABCD$ is shown in the figure $P-V$ diagram. Which of the following curves represent the same processView Solution
- 5Helium at ${27^o}C$ has a volume of $8$ litres. It is suddenly compressed to a volume of $1$ litre. The temperature of the gas will be ....... $^oC$ $[\gamma = 5/3]$View Solution
- 6View SolutionFor an isometric process
- 7A monoatomic gas $\left( {\gamma = \frac{5}{3}} \right)$ is suddenly compressed to $\frac{1}{8}$ of its original volume, then the pressure of gas will change to how many times the initial pressure?View Solution
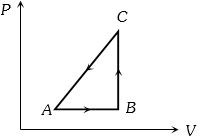
- 8The $P-V$ diagram of a system undergoing thermodynamic transformation is shown in figure. The work done on the system in going from $A → B → C$ is $50 J$ and $ 20\, cal$ heat is given to the system. The change in internal energy between $A$ and $C$ is ...... $J$View Solution
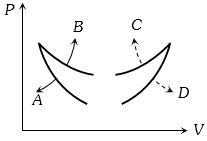
- 9Four curves $A, B, C$ and $D$ are drawn in the adjoining figure for a given amount of gas. The curves which represent adiabatic and isothermal changes areView Solution
- 10During the adiabatic expansion of $2$ moles of a gas, the internal energy of the gas is found to decrease by $2$ joules, the work done during the process on the gas will be equal to ....... $J$View Solution
