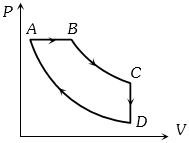
A cyclic process $ABCD$ is shown in the figure $P-V$ diagram. Which of the following curves represent the same process

Medium
(a) $AB$ is isobaric process, $BC$ is isothermal process, $CD$ is isometric process and $DA$ is isothermal process
These process are correctly represented by graph $ (a).$
These process are correctly represented by graph $ (a).$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1If $Q , E$ and $W$ denote respectively the heat added, change in internal energy and the work done by a closed cycle process, thenView Solution
- 2View SolutionThe work done in which of the following processes is zero
- 3Two Carnot engines $A$ and $B$ are operated in succession. The first one, $A$ receives heat from a source at ${T_1} = 800K$ and rejects to sink at ${T_2}K.$. The second engine $B$ receives heat rejected by the first engine and rejects to another sink at ${T_3} = 300K.$ If the work outputs of two engines are equal, then the value of ${T_2}$ is .... $K$View Solution
- 4The value of $\eta$ may lie betweenView Solution
- 5View SolutionWhen an ideal diatomic gas is heated at constant pressure, the fraction of the heat energy supplied which increases the internal energy of the gas, is
- 6Two Carnot engines $A$ and $B$ are operated in succession. The first one, $A$ receives heat from a source at $T_1 = 800\, K$ and rejects to sink at $T_2K$. The second engine $B$ receives heat rejected by the first engine and rejects to another sink at $T_3 = 300\, K$. If the work outputs of two engines are equal, then the value of $T_2$ is ...... $K$View Solution
- 7The volume of air increases by $5\%$ in its adiabatic expansion. The percentage decrease in its pressure will be ...... $\%$View Solution
- 8The work of $146\ kJ$ is performed in order to compress one kilo mole of gas adiabatically and in this process the temperature of the gas increases by $7^o C$. The gas is $(R=8.3\ J\ mol^{-1} K^{-1})$View Solution
- 9View SolutionFor which combination of working temperatures the efficiency of Carnot’s engine is highest
- 10A Car not engine whose low temperate reservoir is at $7\,^oC$ has an efficiency of $50\%$ . It is desired to increase the efficiency to $70\%$ . By how many degrees should the temperature of the high temperature reservoir be increased .... $K$View Solution
