Helium at ${27^o}C$ has a volume of $8$ litres. It is suddenly compressed to a volume of $1$ litre. The temperature of the gas will be ....... $^oC$ $[\gamma = 5/3]$
Medium
(d) $T{V^{\gamma - 1}} = $constant$ \Rightarrow {T_2} = {T_1}{\left( {\frac{{{V_1}}}{{{V_2}}}} \right)^{\gamma - 1}} = {927^o}C$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionCofficient of performance of refigerator is
- 2If $\Delta U$ and $\Delta W$ represent the increase in internal energy and work done by the system respectively in a thermodynamical process, which of the following is true?View Solution
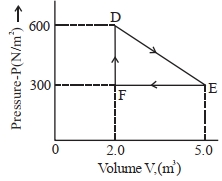
- 3A thermodynamic system is taken from an original state $D$ to an intermediate state $E$ by the linear process shown in the figure. Its volume is then reduced to the original volume from $E$ to $F$ by an isobaric process. The total work done by the gas from $D$ to $E$ to $F$ will be $......J$View Solution
- 4A Carnot engine working between $300\,K$ and $600\,K$ has work output of $800\, J$ per cycle. What is amount of heat energy supplied to the engine from source per cycle...... $J/cycle$View Solution
- 5View SolutionWhen an ideal diatomic gas is heated at constant pressure, the fraction of the heat energy supplied which increases the internal energy of the gas, is
- 6$N _{2}$ gas is heated from $300\, K$ temperature to $600\, K$ through an isobaric process. Then find the change in entropy of the gas. $( n =1 mole )$ (in $J/K$)View Solution
- 7In an isothermal reversible expansion, if the volume of $96\, gm$ of oxygen at $27°C$ is increased from $70$ litres to $140$ litres, then the work done by the gas will beView Solution
- 8An electric heater supplies heat to a system at a rate of $100\;W.$ If system performs work at a rate of $75$ Joules per second. At what rate (in $J/s$) is the internal energy increasing?View Solution
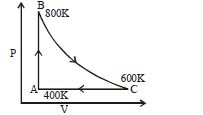
- 9One mole of diatomic ideal gas undergoes a cyclic process $ABC$ as shown in figure. The process $BC$ is adiabatic. The temperatures at $A, B$ and $C$ are $400\,K, 800\,K$ and $600\,K$ respectively. Choose the correct statementView Solution
- 10$\Delta U + \Delta W = 0$ is valid forView Solution
