A black body of surface area $10cm^2$ is heated to $127°C$ and is suspended in a room at temperature $27°C$ . The initial rate of loss of heat from the body at the room temperature will be ...... $W$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Two sheets of thickness $d$ and $3d$, are touching each other. The temperature just outside the thinner sheet side is $A$, and on the side of the thicker sheet is $C$. The interface temperature is $B. A, B$ and $C$ are in arithmetic progressing, the ratio of thermal conductivity of thinner sheet and thicker sheet isView Solution
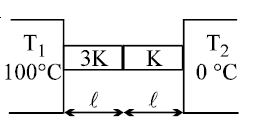
- 2The heat is flowing through two cylindrical rods of same material. The diameters of the rods are in the ratio $1 : 2$ and their lengths are in the ratio $2 : 1$ . If the temperature difference between their ends is the same, the ratio of rate of flow of heat through them will beView Solution
- 3View SolutionMode of transmission of heat, in which heat is carried by the moving particles, is
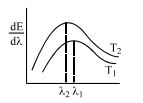
- 4The spectral emissive power $E_\lambda $ for a body at temperature $T_1$ is plotted against the wavelength and area under the curve is found to be $A$. At a different temperature $T_2$ the area is found to be $9A$. Then $\lambda _1/\lambda _2 =$View Solution
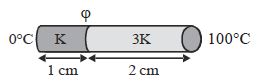
- 5Two bars of thermal conductivities $K$ and $3K$ and lengths $1\,\, cm$ and $2\,\, cm$ respectively have equal cross-sectional area, they are joined lengths wise as shown in the figure. If the temperature at the ends of this composite bar is $0\,^oC$ and $100\,^oC$ respectively (see figure), then the temperature $\varphi $ of the interface is......... $^oC$View Solution
- 6The area of the hole of heat furnace is $10^{-4}\,m^2.$ It radiates $1.58\times 10^5\,cal$ of heat per hour. If the emissivity of the furnace is $0.80,$ then its temperature is .......... $K$View Solution
- 7View SolutionIn rainy season, on a clear night the black seat of a bicycle becomes wet because
- 8Two identical objects $A$ and $B$ are at temperatures $T_A$ and $T_B$ respectively. Both objects are placed in a room with perfectly absorbing walls maintained at temperatures T $({T_A} > T > {T_B}).$ The objects A and B attain temperature $T $ eventually which one of the following is correct statementView Solution
- 9A container with $1 kg$ of water in it is kept in sunlight, which causes the water to get warmer than the surroundings. The average energy per unit time per unit area received due to the sunlight is $700 Wm ^{-2}$ and it is absorbed by the water over an effective area of $0.05 m ^2$. Assuming that the heat loss from the water to the surroundings is governed by Newton's law of cooling, the difference (in ${ }^{\circ} C$ ) in the temperature of water and the surroundings after a long time will be. . . . . . . . (Ignore effect of the container, and take constant for Newton's law of cooling $=0.001 s ^{-1}$, Heat capacity of water $\left.=4200 J kg ^{-1} K ^{-1}\right)$View Solution
- 10View SolutionThe amount of radiation emitted by a perfectly black body is proportional to
