A Car not engine whose low temperate reservoir is at $7\,^oC$ has an efficiency of $50\%$ . It is desired to increase the efficiency to $70\%$ . By how many degrees should the temperature of the high temperature reservoir be increased .... $K$
Diffcult
Initially $\eta=\frac{\mathrm{T}_{1}-\mathrm{T}_{2}}{\mathrm{T}_{1}} \Rightarrow 0.5=\frac{\mathrm{T}_{1}-(273+7)}{\mathrm{T}_{1}}$
$\Rightarrow \frac{1}{2}=\frac{\mathrm{T}_{1}-280}{\mathrm{T}_{1}} \Rightarrow \mathrm{T}_{1}=560 \mathrm{K}$
Finally $\eta_{1}^{\prime}=\frac{T_{1}^{\prime}-T_{2}}{T_{1}^{\prime}} \Rightarrow 0.7=\frac{T_{1}^{\prime}-(273+7)}{T_{1}^{\prime}}$
$\Rightarrow \mathrm{T}_{1}^{\prime}=933 \mathrm{K}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1In thermodynamic process, $200$ Joules of heat is given to a gas and $100$ Joules of work is also done on it. The change in internal energy of the gas is ........ $J$View Solution
- 2View SolutionAn enclosed ideal gas is taken through a cycle as shown in the figure. Then
- 3View SolutionWhich is incorrect
- 4A household refrigerator with a coefficient of performance $1.2$ removes heat from the refrigerated space at the rate of $60\ kJ/min$ .What would be cost of running this fridge for one month $\mathrm{Rs.}$ ..................... $(30\ days)$ (assuming each day it is used for $4$ hours and cost of one electrical unit is $6$ Rs.)View Solution
- 5A refrigerator is to maintain eatables kept inside at $9^{\circ} C .$ If room temperature is $36^{\circ} C$ calculate the coefficient of performance.View Solution
- 6A cylinder fitted with a piston contains $0.2 \,moles$ of air at temperature $27°C.$ The piston is pushed so slowly that the air within the cylinder remains in thermal equilibrium with the surroundings. Find the approximate work done by the system if the final volume is twice the initial volume ...... $J$View Solution
- 7A carnot engine is made to work between $200\,^oC$ and $0\,^oC$ first and then between $0\,^oC$ and $-200\,^oC$. The ratio of efficiencies $\left( {\frac{{{\eta _2}}}{{{\eta _1}}}} \right)$ of the engine in two cases isView Solution
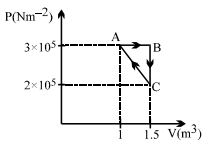
- 8Considere the thermodynamics cycle shown on $PV$ diagram. The process $A \rightarrow B$ is isobaric, $B \rightarrow C$ is isochoric and $C \rightarrow A$ is a straight line process. The following internal energy and heat are given $: \Delta U_{A \rightarrow B} = + 400\,\, kJ$ and $Q_{B \rightarrow C} = - 500\,\, kJ$ The heat flow in the process $Q_{C \rightarrow A}$ is ...... $kJ$View Solution
- 9An engine has an efficiency of $1/6.$ When the temperature of sink is reduced by $62^o C,$ its efficiency is doubled. Temperatures of the source is ....... $^oC$View Solution
- 10View SolutionThe specific heat of a gas at constant pressure is more than that of the same gas at constant volume because
