where $T_1$ is the temperature of the source and $T_2$ is the temperature of the sink.
$\therefore \frac{1}{6} = 1 - \frac{{{T_2}}}{{{T_1}}}\,\,\,or,\,\,\,\frac{{{T_2}}}{{{T_1}}} = \frac{5}{6}$ $...(i)$
where the temperature of the sink is decreased by ${62^ \circ }C\,\left( {or\,62\,K} \right),$ efficiency becomes double.
Since, the temperature of the source remains unchanged
$\therefore 2 \times \frac{1}{6} = 1 - \frac{{\left( {{T_2} - 62} \right)}}{{{T_1}}}\,\,or,\,\,\frac{1}{3} = 1 - \frac{{\left( {{T_2} - 62} \right)}}{{{T_1}}}$
$or,\,\frac{2}{3} = \frac{{{T_2} - 62}}{{{T_1}}}\,\,or,\,2{T_1} = 3{T_2} - 186$
$or,\,\,2{T_1} = 3\left[ {\frac{5}{6}} \right]{T_1} - 186$ $[using (i)]$
$\therefore \,\,\left[ {\frac{5}{2} - 2} \right]{T_1} = 186\,\,or,\,\,\frac{{{T_1}}}{2} = 186$
$or,\,\,{T_1} = 372\,K = {99^ \circ }C.$
Download our appand get started for free
Similar Questions
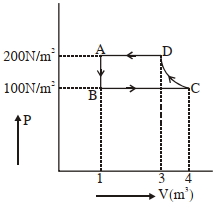
- 1The $P-V$ diagram of a diatomic ideal gas system going under cyclic process as shown in figure. The work done during an adiabatic process $CD$ is (use $\gamma=1.4$) (in $J$)View Solution
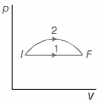
- 2An ideal gas undergoes change in its state from the initial state $I$ to the final state $F$ via two possible paths as shown below. Then,View Solution
- 3During an isothermal expansion, a confined ideal gas does $-150 \,J$ of work against its surroundings. This implies thatView Solution
- 4Carbon monoxide is carried around a closed cycle $abc$ in which bc is an isothermal process as shown in the figure. The gas absorbs $7000 J$ of heat as its temperature increases from $300 K$ to $1000 K$ in going from $a$ to $b$. The quantity of heat rejected by the gas during the process $ca$ is ..... $J$View Solution
- 5Figure below shows two paths that may be taken by a gas to go from a state $A$ to a state $C.$ In process $AB,$ $400 \,J$ of heat is added to the system and in process $BC,$ $100\, J$ of heat is added to the system. The heat absorbed by the system in the process $AC$ will be ...... $J$View Solution
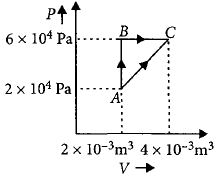
- 6An air bubble of volume $v _0$ is released by a fish at a depth $h$ in a lake. The bubble rises to the surface. Assume constant temperature and standard atmospheric pressure above the lake. The volume of the bubble just before touching the surface will be (density) of water is $\rho$View Solution
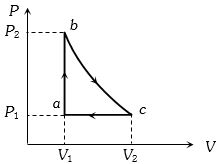
- 7A sample of an ideal gas is taken through the cyclic process $abca$ as shown in the figure. The change in the internal energy of the gas along the path $ca$ is $-180\, J$. The gas absorbs $250\, J$ of heat along the path $ab$ and $60\, J$ along the path $bc$. The work done by the gas along the path $abc$ is ..... $J$View Solution
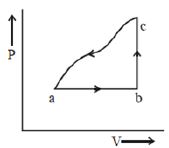
- 8View SolutionAn enclosed ideal gas is taken through a cycle as shown in the figure. Then
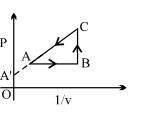
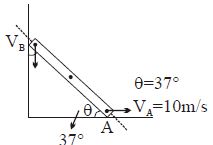
- 9Find $V_B = ?$View Solution
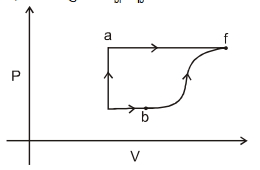
- 10A thermodynamic system is taken form an initial state $i$ with internal energy $U_1=100 \ J$ to the final state along two different paths iaf and ibf, as schematically shown in the fire. The work done by the system along the paths $af$, ib and bf are $W _{ af }=200 \ J , W _{ ID }=50 \ J$ and $W _{ br }=100 \ J$ respectively. The heat supplied to the system along the path iaf, ib and bf are $Q_{\mid a t l} Q_{b r}$ and $Q_{10}$ respectively. If the internal energy of the sytem in the state $b$ is $U_b=$ $200 \ J$ and $Q_{l a t}=500 \ J$, the ratio $Q_{b J} / Q_{10}$ is:View Solution