A carnot engine has the same efficiency between $800 K$ to $500 K$ and $x\, K$ to $600 K.$ The value of $x$ is ...... $K$
Medium
(b) In first case, $({\eta _1}) = 1 - \frac{{500}}{{800}} = \frac{3}{8}$
and in second case, $({\eta _2}) = 1 - \frac{{600}}{x}$
Since ${\eta _1} = {\eta _2},$ therefore $\frac{3}{8} = 1 - \frac{{600}}{x}$
or $\frac{{600}}{x} = 1 - \frac{3}{8} = \frac{5}{8}$or$x = \frac{{600 \times 8}}{5} = 960K$
and in second case, $({\eta _2}) = 1 - \frac{{600}}{x}$
Since ${\eta _1} = {\eta _2},$ therefore $\frac{3}{8} = 1 - \frac{{600}}{x}$
or $\frac{{600}}{x} = 1 - \frac{3}{8} = \frac{5}{8}$or$x = \frac{{600 \times 8}}{5} = 960K$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
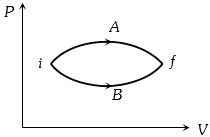
- 1In the figure given two processes $A$ and $B$ are shown by which a thermo-dynamical system goes from initial to final state $F.$ If $\Delta {Q_A}$ and $\Delta {Q_B}$ are respectively the heats supplied to the systems thenView Solution
- 2The pressure and density of a diatomic gas $(\gamma = 7/5)$ change adiabatically from $(P, d)$ to $(P', d')$. If $\frac{{d'}}{d} = 32$, then $\frac{{P'}}{P}$ should beView Solution
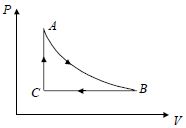
- 3A fixed amount of a gas undergoes a thermodynamic process as shown such that heat interaction along path $B \to C \to A$ is equal to the work done by the gas along path $A \to B \to C$. Then process $A \to B$ is :-View Solution
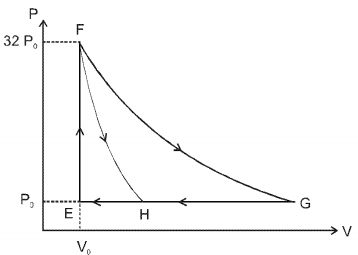
- 4One mole of a monatomic ideal gas is taken along two cyclic processes $E \rightarrow F \rightarrow G \rightarrow E$ and $E \rightarrow F \rightarrow H \rightarrow$ E as shown in the $PV$ diagram. The processes involved are purely isochoric, isobaric, isothermal or adiabatic. $Image$View Solution
Match the paths in List $I$ with the magnitudes of the work done in List $II$ and select the correct answer using the codes given below the lists.
List $I$ List $I$ $P.$ $\quad G \rightarrow E$ $1.$ $\quad 160 P_0 V_0 \ln 2$ $Q.$ $\quad G \rightarrow H$ $2.$ $\quad 36 P _0 V _0$ $R.$ $\quad F \rightarrow H$ $3.$ $\quad 24 P _0 V _0$ $S.$ $\quad F \rightarrow G$ $4.$ $\quad 31 P_0 V_0$ Codes: $ \quad \quad P \quad Q \quad R \quad S $
- 5A carnot engine with its cold body at $17\,^oC$ has $50\%$ effficiency. If the temperature of its hot body is now increased by $145\,^oC$, the efficiency becomes...... $\%$View Solution
- 6View SolutionFirst law thermodynamics states that
- 7View SolutionWhich of the following is not thermodynamical function
- 8Two identical samples of a gas are allowed to expand $(i)$ isothermally $(ii)$ adiabatically. Work done isView Solution
- 9A monoatomic gas at pressure $P$ and volume $V$ is suddenly compressed to one eighth of its original volume. The final pressure at constant entropy will be $.....P$View Solution
- 10$Assertion :$ Thermodynamic process in nature are irreversible.View Solution
$Reason :$ Dissipative effects cannot be eliminated.
