A carnot engine with its cold body at $17\,^oC$ has $50\%$ effficiency. If the temperature of its hot body is now increased by $145\,^oC$, the efficiency becomes...... $\%$
Medium
$\eta=\frac{T_{1}-T_{2}}{T_{1}}$
$\left.\mathrm{T}_{1}=\text { temperature of source (in } \mathrm{K}\right)$
$\mathrm{T}_{2}=$ temperature of $\operatorname{sink}(\mathrm{in} \mathrm{K})$
$\frac{1}{2}=\frac{\mathrm{T}_{1}-290}{\mathrm{T}_{1}}$
$\mathrm{T}_{1}=580 \mathrm{K}$
If $\mathrm{T}_{1}$ is increased by $145^{\circ} \mathrm{C}$
$\mathrm{T}_{1}^{\prime}=580+145=725 \mathrm{K}$
$\eta^{\prime}=\frac{725-290}{725}=0.6$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
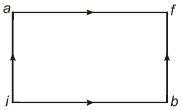
- 1When a system is taken from state $i$ to state $f$ along the path $iaf$, it is found that $Q=50$ $cal$ and $W=20$ $cal$ Along the path $ibf\ Q = 36\ cal. \ W$ along the path $ibf$ is ....... $ cal$View Solution
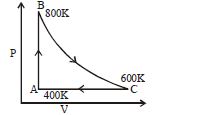
- 2One mole of diatomic ideal gas undergoes a cyclic process $ABC$ as shown in figure. The process $BC$ is adiabatic. The temperatures at $A, B$ and $C$ are $400\,K, 800\,K$ and $600\,K$ respectively. Choose the correct statementView Solution
- 3In case of an adiabatic process the correct relation in terms of pressure $p$ and density $\rho $ of a gas isView Solution
- 4View SolutionAir in a cylinder is suddenly compressed by a piston, which is then maintained at the same position. With the passage of time
- 5The temperature inside and outside of refrigerator are $260\, K$ and $315\, K$ respectively. Assuming that the refrigerator cycle is reversible, calculate the heat delivered to surroundings for every joule of work done.View Solution
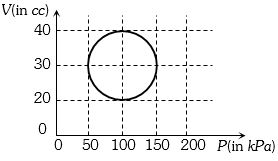
- 6View SolutionA system is taken through a cyclic process represented by a circle as shown. The heat absorbed by the system is
- 7View SolutionFirst law thermodynamics states that
- 8The temperature inside a refrigerator is $t_2 \,^o C$ and the room temperature is $t_1\,^o C.$ The amount of heat delivered to the room for each joule of electrical energy consumed ideally will beView Solution
- 9A gas undergoes a change of state during which $100 J$ of heat is supplied to it and it does $20 J$ of work. The system is brought back to its original state through a process during which $20 J$ of heat is released by the gas. The work done by the gas in the second process is ....... $J$View Solution
- 10View SolutionIn the case of diatomic gas, the heat given at constant pressure is that part of energy which is used for the expansion of gas, is
