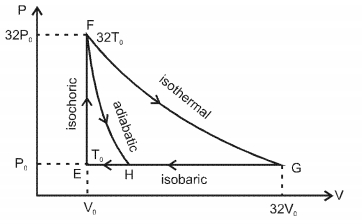
Match the paths in List $I$ with the magnitudes of the work done in List $II$ and select the correct answer using the codes given below the lists.
| List $I$ | List $I$ |
| $P.$ $\quad G \rightarrow E$ | $1.$ $\quad 160 P_0 V_0 \ln 2$ |
| $Q.$ $\quad G \rightarrow H$ | $2.$ $\quad 36 P _0 V _0$ |
| $R.$ $\quad F \rightarrow H$ | $3.$ $\quad 24 P _0 V _0$ |
| $S.$ $\quad F \rightarrow G$ | $4.$ $\quad 31 P_0 V_0$ |
Codes: $ \quad \quad P \quad Q \quad R \quad S $ 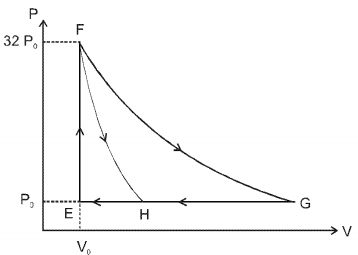
Download our appand get started for free
Similar Questions
- 1A Carnot engine whose low temperature reservoir is at $7\,°C$ has an efficiency of $50\%$. It is desired to increase the efficiency to $70\%$. By how many degrees should the temperature of the high temperature reservoir be increased ....... $K$View Solution
- 2View SolutionA container that suits the occurrence of an isothermal process should be made of
- 3A Carnot engine has efficiency of $50 \%$. If the temperature of sink is reduced by $40^{\circ} C$, its efficiency increases by $30 \%$. The temperature of the source will be$....K$View Solution
- 4Carnot heat engine works with an ideal diatomic gas and an adiabatic volume expansion ratio $32$ . Then its efficiency is ....... $\%$View Solution
- 5During an adiabatic process, the pressure of a gas is found to be proportional to the cube of its temperature. The ratio of $\frac{{{C_P}}}{{{C_V}}}$ for the gas isView Solution
- 6View SolutionIf a system undergoes contraction of volume then the work done by the system will be
- 7View SolutionFor an ideal gas, in an isothermal process
- 8The change in the entropy of a $1$ mole of an ideal gas which went through an isothermal process from an initial state $(P_1, V_1,T)$ to the final state $(P_2, V_2,T)$ is equal toView Solution
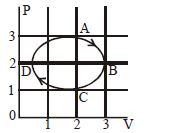
- 9The figure shows the $P-V$ plot of an ideal gas taken through a cycle $ABCDA.$ The part $ABC$ is a semicircle and $CDA$ is half of an ellipse. Then,View Solution
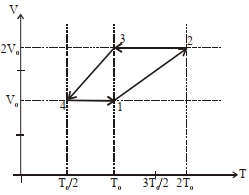
- 10One mole of a monoatomic ideal gas goes through a thermodynamic cycle, as shown in the volume versus temperature ($V-T$) diagram. The correct statement($s$) is/are :View Solution
[ $R$ is the gas constant]
$(1)$ Work done in this thermodynamic cycle $(1 \rightarrow 2 \rightarrow 3 \rightarrow 4 \rightarrow 1)$ is $| W |=\frac{1}{2} RT _0$
$(2)$ The ratio of heat transfer during processes $1 \rightarrow 2$ and $2 \rightarrow 3$ is $\left|\frac{ Q _{1 \rightarrow 2}}{ Q _{2 \rightarrow 3}}\right|=\frac{5}{3}$
$(3)$ The above thermodynamic cycle exhibits only isochoric and adiabatic processes.
$(4)$ The ratio of heat transfer during processes $1 \rightarrow 2$ and $3 \rightarrow 4$ is $\left|\frac{Q_{U \rightarrow 2}}{Q_{3 \rightarrow 4}}\right|=\frac{1}{2}$