A Carnot engine, having an efficiency of $\eta = 1/10$ as heat engine, is used as a refrigerator. If the work done on the system is $10\ J$, the amount of energy absorbed from the reservoir at lower temperature is ....... $J$
AIEEE 2007,NEET 2017,AIPMT 2015,JEE MAIN 2020, Diffcult
The efficiency $\left( \eta \right)$ of a Carnot engine and the coefficient of performance $\left( \beta \right)$ of a refrigerator are related as
$\beta = \frac{{1 - \eta }}{\eta }$ $Here,\,\eta = \frac{1}{{10}}$ $\therefore \beta \frac{{1 - \frac{1}{{10}}}}{{\left( {\frac{1}{{10}}} \right)}} = 9.$
Also, Coefficient of performance $\left( \beta \right)$ is given by $\beta = \frac{{{Q_2}}}{W}$
where $Q_2$ is the energy absorbed from the reservoir.
or, $9 = \frac{{{Q_2}}}{{10}}$ $\therefore {Q_2} = 90\,J$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
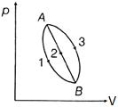
- 1An ideal gas of mass $m$ in a state $A$ goes to another state $B$ via three different processes as shown in figure. If $Q_{1}, Q_{2}$ and $Q_{3}$ denote the heat absorbed by the gas along the three paths, thenView Solution
- 2A Carnot engine whose sink is at $300 \,K$ has an efficiency of $50 \%$. By how much should the temperature of source be increased so as the efficiency becomes $70 \%$ is ............ $K$View Solution
- 3A gas expands adiabatically at constant pressure such that its temperature $T \propto \frac{1}{{\sqrt V }}$, the value of ${C_P}/{C_V}$ of gas isView Solution
- 4View SolutionIn an isothermal process the volume of an ideal gas is halved. One can say that
- 5This question has Statement $1$ and Statement $2.$ Of the four choices given after the Statements, choose the one that best describes the two Statements.View Solution
Statement $1:$ In an adiabatic process, change in internal energy of a gas is equal to work done on/by the gas in the process.Statement $2 :$ The temperature of a gas remains constant in an adiabatic process.
- 6The efficiency of Carnot's engine operating between reservoirs, maintained at temperatures ${27^o}C$ and $ - {123^o}C,$ is ...... $\%$View Solution
- 7View SolutionIn isothermal expansion, the pressure is determined by
- 8Work done by air when it expands from $50\, litres$ to $150\, litres$ at a constant pressure of $2$ atmosphere isView Solution
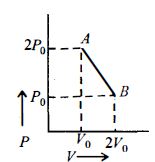
- 9$n\, moles$ of an ideal gas undergo a process $A \to B$ as shown in the figure. Maximum temperature of the gas during the process isView Solution
- 10View SolutionThe door of a working refrigerator is left open in a well insulated room. The temperature of air in the room will
