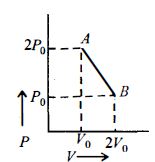
$n\, moles$ of an ideal gas undergo a process $A \to B$ as shown in the figure. Maximum temperature of the gas during the process is

AIEEE 2012, Diffcult
For given graph, equation of $P-V$ line is
$P-2 P_{0}=\frac{2 P_{0}-P_{0}}{V_{0}-2 V_{0}}\left(V-V_{0}\right)$
So, $P=3 P_{0}-\frac{P_{0}}{V_{0}} V$ as $P V=n R T$
$\Rightarrow\left(3 P_{0}-\frac{P_{0}}{V_{0}} \cdot V\right) V=n R T$
For maximum temperature $\frac{d T}{d V}=0$ $\Rightarrow \frac{d T}{d V}=3 P_{0}-\frac{2 P_{0}}{V_{0}} V=0 \Rightarrow V=\frac{3}{2} V_{0}$
Also $P=\frac{3}{2} P_{0}$
So, $T=\frac{P V}{n R}=\frac{1}{n R} \times \frac{3}{2} P_{0} \times \frac{3}{2} V_{0}=\frac{9 P_{0} V_{0}}{4 n R}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionA gas is compressed adiabatically till its temperature is doubled. The ratio of its final volume to initial volume will be
- 2For adiabatic processes $\left( {\gamma = \frac{{{C_p}}}{{{C_v}}}} \right)$View Solution
- 3A diatomic gas with rigid molecules does $10\, J$ of work when expanded at constant pressure. What would be the heat energy absorbed by the gas, in this process ..... $J$.View Solution
- 4A Carnot engine takes $3 \times {10^6}\,cal$. of heat from a reservoir at $627°C$, and gives it to a sink at $27°C.$ The work done by the engine isView Solution
- 5View SolutionHeat is not being exchanged in a body. If its internal energy is increased, then
- 6View SolutionIn practice, all heat engines have efficiency less than that of a Carnot engine because
- 7An air bubble of volume $v _0$ is released by a fish at a depth $h$ in a lake. The bubble rises to the surface. Assume constant temperature and standard atmospheric pressure above the lake. The volume of the bubble just before touching the surface will be (density) of water is $\rho$View Solution
- 8$1 \,\,kg$ of a gas does $20\,\, kJ$ of work and receives $16 \,\,kJ$ of heat when it is expanded between two states. $A$ second kind of expansion can be found between the initial and final state which requires a heat input of $9\,\, kJ$. The work done by the gas in the second expansion is ....... $kJ$View Solution
- 9One mole of helium is adiabatically expanded from its initial state $({P_i},{V_i},{T_i})$ to its final state $({P_f},{V_f},{T_f})$. The decrease in the internal energy associated with this expansion is equal toView Solution
- 10A bicycle tyre is filled with air having pressure of $270\,kPa$ at $27^{\circ}\,C$. The approximate pressure of the air in the tyre when the temperature increases to $36^{\circ}\,C$ is $............kPa$View Solution
