Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A Carnot engine absorbs $1000\,J$ of heat energy from a reservoir at $127\,^oC$ and rejects $600\,J$ of heat energy during each cycle. The efficiency of engine and temperature of sink will beView Solution
- 2View SolutionWhich of the following statements is correct for any thermodynamic system
- 3If one mole of an ideal gas goes through the process $A \rightarrow B$ and $B \rightarrow C .$ Given that $T _{ A }=400\, K ,$ and $T _{ C }=400 \,K .$ If $\frac{ P _{ B }}{ P _{ A }}=\frac{1}{5},$ then find the heat supplied to the gas (in $J$)View Solution
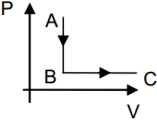
- 4A monoatomic idea gas expands at constant pressure, with heat $Q$ supplied. The fraction of $Q$ which goes as work done by the gas isView Solution
- 5The pressure in the tyre of a car is four times the atmospheric pressure at $300 K$. If this tyre suddenly bursts, its new temperature will be $(\gamma = 1.4)$View Solution
- 6During the melting of a slab of ice at $273\, K$ at atmospheric pressureView Solution
- 7The temperature inside and outside a refrigerator are $273 \,K$ and $300 \,K$ respectively. Assuming that the refrigerator cycle is reversible, for every joule of work done, the heat delivered to the surrounding will be nearly ........ $J$View Solution
- 8Considere the thermodynamics cycle shown on $PV$ diagram. The process $A \rightarrow B$ is isobaric, $B \rightarrow C$ is isochoric and $C \rightarrow A$ is a straight line process. The following internal energy and heat are given $: \Delta U_{A \rightarrow B} = + 400\,\, kJ$ and $Q_{B \rightarrow C} = - 500\,\, kJ$ The heat flow in the process $Q_{C \rightarrow A}$ is ...... $kJ$View Solution
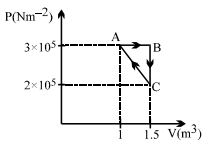
- 9If one mole of an ideal gas at $\left( P _{1}, V _{1}\right)$ is allowed to expand reversibly and isothermally ($A$ to $B$ ) its pressure is reduced to one-half of the original pressure (see $figure$). This is followed by a constant volume cooling till its pressure is reduced to one-fourth of the initial value $( B \rightarrow C ) .$ Then it is restored to its initial state by a reversible adiabatic compression ($C$ to $A$). The net workdone by the gas is equal to ...... .View Solution
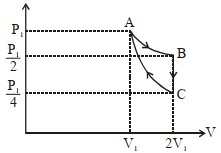
- 10$A$ reversible engine converts one-sixth of the heat input into work. When the temperature of the sink is reduced by $ 62^oC$, the efficiency of the engine is doubled. The temperatures of the source and sink areView Solution
