A Carnot engine absorbs $1000\,J$ of heat energy from a reservoir at $127\,^oC$ and rejects $600\,J$ of heat energy during each cycle. The efficiency of engine and temperature of sink will be
JEE MAIN 2014, Diffcult
Given: $Q_1=1000 J$
$Q_2=600 J$
${T_1} = {127^ \circ }C = 400\,K$
${T_2} = ?$
$\eta = ?$
Efficiency of carnot engine,
$\eta = \frac{W}{Q_1} \times 100\% $
$or,\,\,\eta = \frac{{{Q_2} - {Q_1}}}{{{Q_1}}} \times 100\% $
$or,\,\,\,\eta = \frac{{1000 - 600}}{{1000}} \times 100\% $
$\eta = 40\% $
$Now,for\,carnot\,cycle\frac{{{Q_2}}}{{{Q_1}}} = \frac{{{T_2}}}{{{T_1}}}$
$\frac{{600}}{{1000}} = \frac{{{T_2}}}{{400}}$
${T_2} = \frac{{600 \times 400}}{{1000}} = 240\,K = 240 - 273$
$\therefore {T_2} = - {33^ \circ }C$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
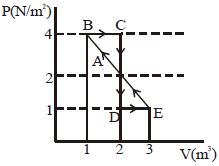
- 1One mole of a monoatomic gas is carried along process $ABCDEA$ as shown in diagram. Find the net work done by gas ......... $J$View Solution
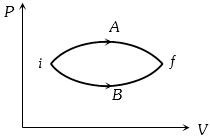
- 2In the figure given two processes $A$ and $B$ are shown by which a thermo-dynamical system goes from initial to final state $F.$ If $\Delta {Q_A}$ and $\Delta {Q_B}$ are respectively the heats supplied to the systems thenView Solution
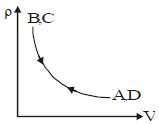
- 3Density $vs$ volume graph is shown in the figure. Find corresponding pressure $vs$ temperature graphView Solution
- 4Three samples of the same gas $A, B$ and $C(\gamma = 3/2)$ have initially equal volume. Now the volume of each sample is doubled. The process is adiabatic for $A$ isobaric for $B $ and isothermal for $C$. If the final pressures are equal for all three samples, the ratio of their initial pressures areView Solution
- 5Jet aircrafts fly at altitudes above $30000 \,ft$, where the air is very cold at $-40^{\circ} C$ and the pressure is $0.28 \,atm$. The cabin is maintained at $1 \,atm$ pressure by means of a compressor which exchanges air from outside adiabatically. In order to have a comfortable cabin temperature of $25^{\circ} C$, we will require in additionView Solution
- 6Column $I$ Contains a list of processes involving expansion of an ideal gas. Match this with Column $II$ describing the thermodynamic change during this process. Indicate your answer by darkening the appropriate bubbles of the $4 \times 4$ matrix given in the $ORS$.View Solution
Column $I$ Column $II$ $(A)$ An insulated container has two chambers separated by a valve. Chamber $I$ contains an ideal gas and the Chamber $II$ has vacuum. The valve is opened. $(p)$ The temperature of the gas decreases $(B)$ An ideal monoatomic gas expands to twice its original volume such that its pressure $\mathrm{P} \propto \frac{1}{\mathrm{~V}^2}$, where $\mathrm{V}$ is the volume of the gas $(q)$ The temperature of the gas increases or remains constant $(C)$ An ideal monoatomic gas expands to twice its original volume such that its pressure $\mathrm{P} \propto \frac{1}{\mathrm{~V}^{4 / 3}}$, where $\mathrm{V}$ is its volume $(r)$ The gas loses heat $(D)$ An ideal monoatomic gas expands such that its pressure $\mathrm{P}$ and volume $\mathrm{V}$ follows the behaviour shown in the graph $Image$ $(s)$ The gas gains heat 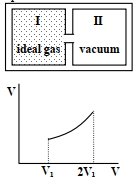
- 7$N _{2}$ gas is heated from $300\, K$ temperature to $600\, K$ through an isobaric process. Then find the change in entropy of the gas. $( n =1 mole )$ (in $J/K$)View Solution
- 8View SolutionA gas is compressed isothermally to half its initial volume. The same gas is compressed separately through an adiabatic process until its volume is again reduced to half. Then
- 9A monatomic gas at pressure $P_1$ and volume $V_1$ is compressed adiabatically to ${\frac{1}{8}}^{th}$ of its original volume. What is the final pressure of the gas is ........ $P_1$?View Solution
- 10A sample of $0.1\, g$ of water at $100^o C$ and normal pressure $(1.013 \times 10^5 N m^{-2} )$ requires $54\ cal $ of heat energy to convert to steam at $100^o C.$ If the volume of the steam produced is $167.1 \,cc,$ the change in internal energy of the sample, is ....... $J$View Solution
