Which of the following statements is correct for any thermodynamic system
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionWhich of the following is a slow process
- 2A diatomic gas, having $C_{p}=\frac{7}{2} R$ and $C _{ v }=\frac{5}{2} R ,$ is heated at constant pressure. The ratio $dU : dQ : dW :$View Solution
- 3An ideal Carnot heat engine with an efficiency of $30\%$.It absorbs heat from a hot reservoir at $727^o C$. The temperature of the cold reservoir is .... $^oC$View Solution
- 4The temperature inside a refrigerator is $t_2 \,^o C$ and the room temperature is $t_1\,^o C.$ The amount of heat delivered to the room for each joule of electrical energy consumed ideally will beView Solution
- 5At ${27^o}C$ a gas is suddenly compressed such that its pressure becomes $\frac{1}{8}th$ of original pressure. Temperature of the gas will be $(\gamma = 5/3)$View Solution
- 6When heat energy of $1500\; Joules$, is supplied to a gas at constant pressure $2.1 \times {10^5}\;N/{m^2}$, there was an increase in its volume equal to $2.5 \times {10^{ - 3}}\;{m^3}$. The increase in internal energy of the gas in Joules is ...... $J$View Solution
- 7Following figure shows two processes $A$ and $B$ for a gas. If $\Delta Q_A$ and $\Delta Q_B$ are the amount of heat absorbed by the system in two cases, and $\Delta U_A$ and $\Delta U_B$ are changes in internal energies, respectively, thenView Solution
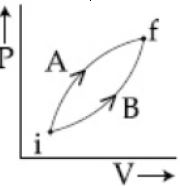
- 8The volume of an ideal gas $(\gamma=1.5)$ is changed adiabatically from $5$ litres to $4$ litres. The ratio of initial pressure to final pressure is:View Solution
- 9An ideal gas undergoes cyclic process as shown in density pressure graph. During the process $AB$ the work done $|W_{AB}| = 70\,J$ . During the process $BC$, the gas absorbs $150\,J$ of heat. During the process $CA$ , gas undergoes expansion and does $210\,J$ of workView Solution
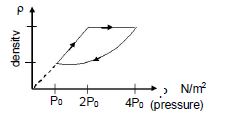
- 10An engine runs between a reservoir at temperature $200 \,K$ and a hot body which is initially at temperature of $600 \,K$. If the hot body cools down to a temperature of $400 \,K$ in the process, then the maximum amount of work that the engine can do (while working in a cycle) is (the heat capacity of the hot body is $1 \,J / K )$View Solution
